issue contents
February 2009 issue
Low-resolution structure determination and validation
Proceedings of the CCP4 study weekend
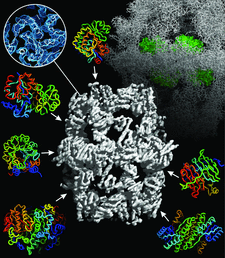
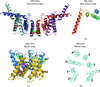
Cover illustration: Crystallographic studies of the fungal fatty acid synthase (FAS) multienzyme (p. 101). Homologous structures of individual catalytic domains were initially fitted into a 5 Å resolution electron-density map. Phase extension to higher resolution allowed building and refinement of the atomic FAS structure shown in the background.
research papers
Open  access
access
 access
accessA case of imperfect pseudo-merohedral twinning in monoclinic crystals of fungal fatty acid synthase is discussed. A space-group transition during crystal dehydration resulted in a Moiré pattern-like interference of the twinned diffraction patterns.
Open  access
access
 access
accessX-ray crystallographic and complementary functional studies have contributed significantly to the current understanding of gene transcription. Here, recent structure–function studies on various aspects of the elongation phase of transcription are summarized.
Open  access
access
 access
accessInterpretation of low-resolution X-ray crystallographic data can prove to be a difficult task. The challenges faced in electron-density interpretation, the strategies that have been employed to overcome them and developments to automate the process are reviewed.
Open  access
access
 access
accessRefinement is meaningful even at 4 Å or lower, but with present methodologies it should start from high-resolution crystal structures whenever possible.
Open  access
access
 access
accessThe need for validation of macromolecular crystal structures is discussed. A general approach to validation is presented, together with examples of its implementation in the special case of macromolecular crystallography.
Open  access
access
 access
accessA case-matched control protocol provides a useful method to compare sets of macromolecular structures.
Open  access
access
 access
accessThis paper reports on the current status of structure validation in chemical crystallography.
Open  access
access
 access
accessThe article summarizes the information that is gained from and the errors that are found in carbohydrate structures in the Protein Data Bank. Validation tools that can locate these errors are described.
Open  access
access
 access
accessIn a first systematic exploration of phasing with Rosetta de novo models, it is shown that all-atom refinement of coarse-grained models significantly improves both the model quality and performance in molecular replacement with the Phaser software.
Open  access
access
 access
accessAn evaluation of validation and real-space intervention possibilities for improving existing automated (re-)refinement methods.
Open  access
access
 access
accessThe ongoing story of the structure of EmrE, the archetypical member of the small multidrug-resistance family, is described.
Open  access
access
 access
accessAn analysis is presented of the methodological errors that led to the incorrect structure of MsbA.


 journal menu
journal menu