issue contents
April 2005 issue
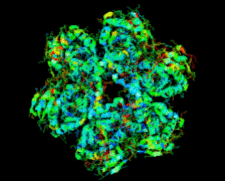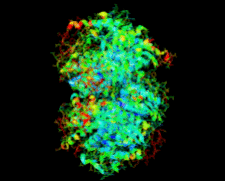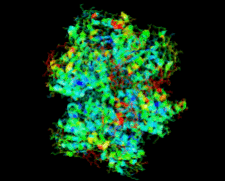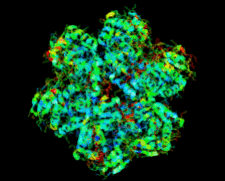
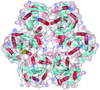
Cover illustration: Salmonella typhimurium uridine phosphorylase (p. 337).
protein structure communications
S. typhimurium uridine phosphorylase is a homohexamer and is structurally homologous to the hexameric purine nucleoside phosphorylases.
PDB reference: StUPh, 1sj9, r1sj9sf
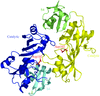
The 1.8 Å crystal structure of an MBP-fusion protein with the C-terminal cytoplasmic segment of the V1 vasopressin receptor reveals that the receptor segment is unstructured.
PDB reference: MBP-V1(Ct), 1ytv, r1ytvsf
A high-resolution structure of human triosephosphate isomerase was obtained from crystals improved by means of the gel-tube method.
PDB reference: triosephosphate isomerase, 1wyi, r1wyisf
structural genomics communications
The crystal structure of H. influenzae RsuA has been determined and refined at 1.59 Å resolution. While the overall structures of the individual S4 and catalytic domains are very similar to those of E. coli RsuA, a large conformational change between these two domains observed in H. influenzae RsuA offers insight into the possible mode of rRNA recognition by this enzyme.
PDB reference: RsuA, 1vio, r1viosf
crystallization communications
Crystals of a non-haemorrhagic fibrin(ogen)olytic metalloproteinase from the venom of A. acutus have been obtained and characterized by X-ray diffraction.
Dihydropyrimidinase from the yeast S. kluyveri was crystallized by vapour diffusion. The crystals belong to space group P21 (unit-cell parameters a = 91.0, b = 73.0, c = 161.4 Å, β = 91.4°) and diffracted to 2.6 Å resolution.
Crystallization and preliminary X-ray analysis of immunophilin-like FKBP42 from Arabidopsis thaliana
The crystallization of FKBP42, a multi-domain member of the FK506-binding protein family, from the plant A. thaliana is reported.
The histidine-containing phosphotransfer protein ZmHP1 from maize was crystallized. The removal of an N-terminal His tag resulted in a remarkable improvement of the diffraction data.
Crystallization and preliminary X-ray diffraction of the ZO-binding domain of human occludin.
The product of the human leukocyte antigen (HLA) gene HLA-B*2703 differs from that of the prototypical subtype HLA-B*2705 by a single amino acid at heavy-chain residue 59 that is involved in anchoring the peptide N-terminus within the A pocket of the molecule. Two B*2703–peptide complexes were crystallized using the hanging-drop vapour-diffusion method using PEG 8000 as a precipitant. A pocket of the molecule, two HLA-B*2703–peptide complexes were crystallized and data sets were collected to high resolution using synchrotron radiation.
The tRNA-specific adenosine deaminase from the pathogenic bacteria S. pyogenes has been overexpressed and crystallized.
The crystallization and preliminary X-ray diffraction analysis of MraZ, formerly known as hypothetical protein YabB, from Escherichia coli K-12 is presented.
The receptor protein tyrosine phosphatase CRYP-2 has been shown to be an inhibitory factor for the growth of retinal axons in the chick. The cloning, expression, purification and crystallization of the catalytic domain of CRYP-2 are reported here.
The protein SH3BGRL, containing both SH3-binding and Homer EVH1-binding motifs, has been crystallized using the hanging-drop vapour-diffusion method.
Thioredoxin reductase 1 (Trr1) from S. cerevisiae is a component of the thioredoxin system, which is involved in several biological processes, including the reduction of disulfide bonds and response to oxidative stress. The expression, purification, crystallization and preliminary X-ray crystallographic studies of yeast Trr1 are reported.
In order to clarify the structural basis of the pathogenesis-related-1 domain, Na-ASP-1, the first multi-domain ASP from the human hookworm parasite N. americanus, has been crystallized. 2.2 Å resolution data have been collected from a crystal belonging to the monoclinic space group P21.
Cytochrome c552 of a moderate thermophile, H. thermoluteolus, was overexpressed in E. coli and crystallized for X-ray diffraction study.
The crystallization and preliminary X-ray analysis of AzoR (azoreductase) have been performed.
Crystals of 3-deoxy-D-arabino-heptulosonate-7-phosphate synthase from M. tuberculosis have been grown and a native data set has been collected to a maximum resolution of 2.5 Å.
Er. carotovora L-asparaginase, a potential antileukaemic agent, has been crystallized. Crystals diffract to 2.6 Å using a rotating-anode source and belong to space group P21, with unit-cell parameters a = 78.0, b = 112.3, c = 78.7 Å, β = 101.9° and a homotetramer in the crystallographic asymmetric unit.
APH(2′′)-Ib is an enzyme responsible for high-level gentamicin resistance in E. faecium isolates. Native crystals of this enzyme have been prepared and preliminary X-ray diffraction experiments have been undertaken.
A plant- and prokaryote-conserved domain (PPC) has been crystallized. The crystal diffracted to 1.7 Å resolution and belonged to space group P6322.
Calcium-binding protein-2 (EhCaBP2) crystals were grown using MPD as a precipitant. EhCaBP2 also crystallized in complex with strontium (replacing calcium) at similar conditions. Preliminary data for EhCaBP2 crystals in complex with an IQ motif are also reported.
Open  access
access
 access
accessDomain III of the West Nile virus envelope protein, the putative receptor-binding domain, was reported to be intrinsically unstable and has defied previous crystallization attempts. It has now been purified from inclusion bodies by protein refolding and was crystallized using the hanging-drop vapour-diffusion method at 291 K.
φ29 bacteriophage scaffolding protein (gp7) has been overproduced in E. coli, purified, crystallized and characterized by X-ray diffraction. Two distinct crystal forms were obtained and a diffraction data set was collected to 1.8 Å resolution.
The periplasmic chaperone FaeE of E. coli F4 fimbriae crystallizes in three crystal forms.
L-Threonine dehydrogenase from the hyperthermophilic archaeon P. horikoshii was crystallized and preliminary X-ray crystallographic analysis was carried out.
The feasibility of glycosylation post-purification has been demonstrated by introducing glucose into the model protein lysozyme via a novel reaction that is compatible with biological samples.
An X-ray crystallographic study of a LOX mutant in which Arg181 is replaced by Met was initiated in order to understand the functions of the conserved amino-acid residues around the FMN in the enzyme active site. LOX-R181M crystals belong to the tetragonal space group I422, with unit-cell parameters a = b = 192.632, c = 200.263 Å, α = β = γ = 90°.
Open  access
access
 access
accessP. aeruginosa CobE, a protein implicated in vitamin B12 biosynthesis, has been crystallized and data on the native and SeMet forms recorded to resolutions of 1.9 and 1.7 Å, respectively. The anomalous measurements will be used for phasing.
Glutaredoxin 2 (Grx2) from S. cerevisiae was expressed, purified and crystallized using the hanging-drop vapour-diffusion method. Crystals were obtained from protein treated with t-butyl hydroperoxide and from a sample not submitted to pre-treatment. Both crystals belong to the tetragonal space group P41212, with very similar unit-cell parameters, and diffraction data were collected to 2.05 and 2.15 Å resolution, respectively.
A truncated form of hemolysin A from P. mirabilis capable of activating hemolytic activity has been cloned, expressed, purified and crystallized.


 journal menu
journal menu







































![[publBio]](http://journals.iucr.org/logos/publbio.gif)





