issue contents
June 2005 issue
Cover illustration: Native histone octamer from chick erythrocytes (p. 541).
protein structure communications
Open  access
access
 access
accessThe high-resolution (1.90 Å) model of the native histone octamer allows structural comparisons to be made with the nucleosome-core particle, along with an identification of a likely core-histone binding site.
PDB reference: histone octamer, 1tzy, r1tzysf
The three-dimensional structure of C. freundii L-methionine γ-lyase has been determined at 1.9 Å resolution.
PDB reference: L-methionine γ-lyase, 1y4i, r1y4isf
crystallization communications
Ectromelia virus glutaredoxin has been crystallized in the presence of the reducing agent DTT. A diffraction data set has been collected and processed to 1.8 Å resolution.
Adenosine kinase from M. tuberculosis has been overexpressed, purified and crystallized in the presence of adenosine. Structure determination using molecular replacement with diffraction data collected at 2.2 Å reveals a dimeric structure.
The production, purification, crystallization and preliminary X-ray crystallographic analysis of adeno-associated virus serotype 8 is reported.
Ervatamin A is a papain-family cysteine protease with high activity and stability. It has been isolated and purified from the latex of the medicinal flowering plant E. coronaria and crystallized by the vapour-diffusion technique. Crystals diffracted to 2.1 Å and the structure was solved by molecular replacement.
Crystals grown by simple dehydration from a solution of hypoxic response protein I (HRPI) from M. tuberculosis diffract to a maximum resolution of 2.1 Å
A. polyphaga mimivirus, the largest known double-stranded DNA virus, is the first virus to exhibit a nucleoside diphosphate kinase gene. The expression and crystallization of the viral NDK are reported.
Preliminary data analysis of crystals of a designed six-finger zinc-finger protein bound to DNA is presented. The cell likely contains two protein–DNA complexes and diffracts to 2.95 Å.
The electron-transfer complex between the terminal oxygenase and ferredoxin of carbazole 1,9a-dioxygenase was crystallized and diffraction data were collected to 1.90 Å resolution.
Open  access
access
 access
accessThe gene encoding the putative mevalonate diphosphate decarboxylase, an enzyme from the mevalonate pathway of isoprenoid precursor biosynthesis, has been cloned from T. brucei. Recombinant protein has been expressed, purified and highly ordered crystals obtained and characterized to aid the structure–function analysis of this enzyme.
A novel oligonucleotide containing cyclohexene-nucleoside building blocks has been crystallized. Crystals belong to space group R3 and diffract to 1.7 Å.
Upon purification, an N-terminally deleted version of the Hermes transposase exists in solution as a mixture of two species that are approximately hexameric and dimeric. Crystals have been obtained of the smaller species that diffract to 2.1 Å resolution.
Nucleoside kinase from the hyperthermophilic archaeon M. jannaschii is a member of the PFK-B family which belongs to the ribokinase superfamily. Here, its expression, purification, crystallization and preliminary X-ray analysis are described.
Crystals of the 39 kDa functional form of methylthioribose-1-phosphate isomerase from B. subtilis diffracted to 2.50 Å.
Crystals of the cytoplasmic domain of FlhA, a membrane-protein component of the bacterial flagellar type III protein-export apparatus from Salmonella, have been obtained and characterized by X-ray diffraction.
The molecular chaperone-like reactivating factor for adenosylcobalamin (coenzyme B12) dependent diol dehydratase was crystallized in ADP-bound and nucleotide-free forms. Preliminary X-ray analysis indicated that crystals are orthorhombic and diffract to 2.0 Å.
Two crystal forms of the antibiotic resistance enzyme APH(9)-Ia from L. pneumophila are reported.
Δ1-Pyrroline-5-carboxylate dehydrogenase from T. thermophilus has been crystallized in a citrate-bound form. The crystals exhibited hemihedral twinning and untwinned crystals were obtained by exclusion of lithium sulfate from the crystallization solution. Apo, NAD+ bound-, NADH bound- and glutamate-bound forms were also obtained and characterized by X-ray diffraction.
Calcineurin B homologous protein 1 from rat was expressed in E. coli, purified and crystallized. A full set of X-ray diffraction data has been collected to 2.2 Å resolution using a synchrotron-radiation source.
Open  access
access
 access
accessCrystallization of the first rhesus macaque MHC class I complex.
The sialic acid-binding domain (VP8*) component of the porcine CRW-8 rotavirus spike protein has been overexpressed in E. coli, purified and co-crystallized with an N-acetylneuraminic acid derivative. X-ray diffraction data have been collected to 2.3 Å, which has enabled determination of the structure by molecular replacement.


 journal menu
journal menu











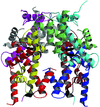





















![[publBio]](http://journals.iucr.org/logos/publbio.gif)





