issue contents
June 2006 issue
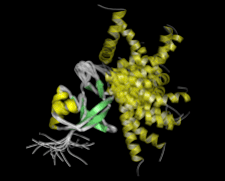
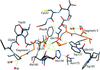
Cover illustration: Solution structure of Arabidopsis thaliana protein At5g39720.1 (p. 490).
editorial
Free 

structural genomics communications
The solution structure of A. thaliana protein At5g39720.1 reported here is the first for a member of the AIG2-like family (PF06094). The three-dimensional structure shows similarity to those determined for members of the uncharacterized Pfam family UPF0131.
PDB reference: At5g39720.1, 2g0q
Open  access
access
 access
accessThe structure of the PII signal transduction protein of N. meningitidis at 1.85 Å resolution is described.
PDB reference: PII signal transduction protein, 2gw8, r2gw8sf
The crystal structure of a conserved hypothetical protein from X. campestris has been determined to a resolution of 1.6 Å. The determined X. campestris structure shows that it belongs to the superfamily of serine α/β hydrolase, with an extra strand preceding the first β-strand to lead to extensive subunit interactions in the crystal.
PDB reference: XC6422, 2fuk
protein structure communications
Open  access
access
 access
accessAttempts to crystallize a complex of papain (C. papaya) with a cysteine protease inhibitor from the parasitic pathogen T. brucei failed. However, over an extended period the mixture produced an ordered crystal of the protease carrying two peptide fragments in the active site. These correspond to dipeptides and tripeptides that are assigned as fragments of the inhibitor, which has presumably suffered proteolytic cleavage.
Data to a maximum of 2.7 Å resolution have allowed a more accurate model of the extracellular domains of the β common cytokine receptor to be determined. Structural details of the membrane-distal BC and FG loops of domain 4 and loops AB and EF of domain 1, which have been implicated in high-affinity binding, are much better defined, revealing a more compact functional epitope.
PDB reference: human β common receptor, 2gys
crystallization communications
The C-terminal protease domain of Venezuelan equine encephalitis virus (VEEV) nsP2 has been overexpressed in E. coli, purified and successfully crystallized. Native crystals diffract to beyond 2.5 Å resolution and isomorphous heavy-atom derivatives suitable for phase analysis have been identified.
Open  access
access
 access
accessThree mutants of the soluble quinoprotein glucose dehydrogenase and two homologues from E. coli and S. coelicolor have been crystallized using an efficient microseeding method.
Rice Bowman–Birk inhibitor was expressed and crystallized.
Sample preparation, characterization, crystallization and preliminary X-ray analysis are reported for a HNF1β–DNA binary complex.
A recombinant subcomplex of the peripheral stalk or stator domain of the ATP synthase from bovine mitochondria has been crystallized and a native data set has been collected to 2.8 Å resolution.
The N-terminal, cytoplasmic domain of the Na+-coupled HCO3 cotransporter NBCe1-A crystallizes in the trigonal space group P3121 or P31 with pseudo P3121 symmetry and diffracts X-rays to 3.0 Å resolution. The crystal packing demonstrates a domain-swap mechanism for dimerization.
Crystallization and X-ray data analyses were successful for both Ca2+-independent and Ca2+-dependent species of the type II antifreeze protein. The resolution of the crystal was 1.35 Å for the Ca2+-independent species, and was 1.25 and 1.06 Å for the Ca2+-dependent species in the Ca2+-free and -bound states, respectively.
Orotidine 5′-monophosphate decarboxylase of human malaria parasite P. falciparum was crystallized by the seeding method in a hanging drop using PEG 3000 as a precipitant. A complete set of diffraction data from a native crystal was collected to 2.7 Å resolution at 100 K using synchrotron radiation.
The purification, crystallization and preliminary structural characterization of human MAWD-binding protein (MAWDBP) are described.
Recombinant L-rhamnose isomerase from P. stutzeri has been crystallized. Diffraction data have been collected to 2.0 Å resolution.
Salicylate 1,2-dioxygenase, a new ring-fission dioxygenase from the naphthalenesulfonate-degrading strain P. salicylatoxidans, which oxidizes salicylate to 2-oxohepta-3,5-dienedioic acid by a novel ring-fission mechanism, has been crystallized. The crystals obtained give diffraction data to 2.9 Å resolution which could assist in the elucidation of the catalytic mechanism of this peculiar dioxygenase.
The immunoglobulin-like domain 3 of human palladin has been crystallized and crystals diffract to 1.8 Å resolution.
In order to investigate tRNA identity elements, an elongator tRNASer acceptor-stem helix was crystallized and a data set was collected to 1.8 Å resolution aiming at a comparison with the corresponding region in suppressor tRNASec.
Antigen-free Fab fragment of mAbR310, which recognizes (R)-HNE modified protein, has been crystallized. Initial phases have been obtained by molecular replacement.
The cytochrome c nitrite reductase complex from D. vulgaris Hildenborough has been crystallized. The preliminary crystallographic structure reveals a 2:1 NrfA:NrfH complex stoichiometry.
The complex of NADH and 3α-HSD from Pseudomonas sp. B-0831 has been crystallized and X-ray diffraction data have been collected to 1.8 Å resolution.
Lysine ∊-aminotransferase from M. tuberculosis has been crystallized. Preliminary crystallographic analysis shows that there is one monomer in the asymmetric unit of the crystal.
Enoyl-acyl carrier protein (ACP) reductases are responsible for bacterial type II fatty-acid biosynthesis and are attractive targets for developing novel antibiotics. The S. pneumoniae enoyl-ACP reductase (FabK) was crystallized and selenomethionine MAD data were collected to 2 Å resolution.
The gene encoding M. tuberculosis FPGS (MtbFPGS; Rv2447c) has been cloned and the protein has been expressed in E. coli. The purified protein was crystallized and X-ray diffraction data were collected to 2.0 Å resolution.
SecA ATPase from E. faecalis has been cloned, overexpressed, purified and crystallized. Crystals belong to space group C2 and diffract to 2.4 Å resolution.
NAD+-preferring aldohexose dehydrogenase from the thermoacidophilic archaeon T. acidophilum was crystallized using the hanging-drop vapour-diffusion technique and X-ray diffraction data were collected to a resolution of 2.8 Å.
BphA3, a Rieske-type [2Fe–2S] ferredoxin, was crystallized by the hanging-drop vapour-diffusion method. A molecular-replacement calculation yielded a satisfactory solution.
The glutathione peroxidase Gpx3 from the yeast S. cerevisiae has been overexpressed, purified, crystallized and diffracted to 2.6 Å resolution. Gpx3 is a monomer in solution which is different from its counterparts in mammals.
The D. melanogaster Drep-3 protein has been crystallized. Crystals were obtained at 293 K that diffracted to 2.8 Å resolution and belonged to space group P212121.
A non-catalytic and myotoxic Lys49-PLA2 from B. jararacussu venom was crystallized with BPB inhibitor and X-ray diffraction data were collected. Preliminary analysis indicates that the ligand is bound to the His48 residue. Structure determination may provide insights into the myotoxic and cytotoxic mechanisms of Lys49-PLA2s.
Open  access
access
 access
accessCrystals of T. gondii ENR in complex with NAD+ and triclosan were grown using the hanging-drop vapour-diffusion method with PEG 8000 as precipitant.
Crystals of the catalytic PRONE domain of the guanine nucleotide exchange factor RopGEF8 and its complex with the Rho-family protein Rop4 from A. thaliana were obtained that diffract to 2.2 and 3.1 Å resolution, respectively.
books received
Free 



 journal menu
journal menu








































![[publBio]](http://journals.iucr.org/logos/publbio.gif)





