issue contents
September 2008 issue
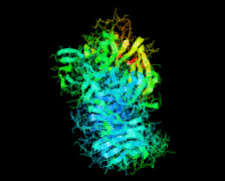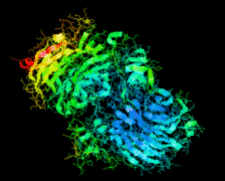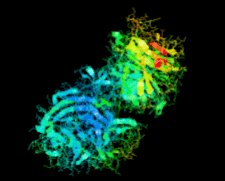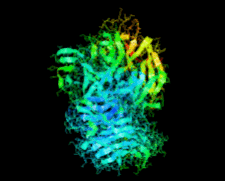
Cover illustration: Catalytic domain of Streptococcus pneumoniae sialidase NanA (Xu et al., p. 772).
protein structure communications
Open  access
access
 access
accessThe structure of a catalytically active subdomain of the NanA sialidase from S. pneumoniae is reported to a resolution of 2.5 Å. The complex with the inhibitor Neu5Ac2en identifies the key catalytic residues and provides a platform for structure-based development of specific inhibitors.
PDB reference: NanA sialidase, 2vvz, r2vvzsf
The structure of the catalytic subunit of M. jannaschii aspartate transcarbamoylase has been determined in space group P212121 using synchrotron data to a resolution of 3.0 Å and was refined to a final Rwork and Rfree of 0.215 and 0.269, respectively.
PDB reference: aspartate transcarbamoylase, 3e2p, r3e2psf
crystallization communications
Acidic nondenaturing PAGE revealed inhomogeneity of the MASP-1 catalytic region caused by deamidation as the reason behind previous unsuccessful crystallization attempts. Monitoring the separation of the various species by acidic nondenaturing PAGE during purification helped to obtain pure protein, which was subsequently crystallized.
Histamine dehydrogenase from Nocardioides simplex has been expressed, purified and crystallized with full incorporation of 6-S-cysteinyl-FMN. Diffraction data have been collected to 2.7 Å resolution; the crystals belonged to the orthorhombic space group P212121.
Prolyl-4-hydroxylase from B. anthracis has been cloned, expressed and crystallized. A complete MAD data set has been collected to 1.4 Å resolution.
Crystallization of the proposed FAD-containing ferri-siderophore reductase protein YqjH from E. coli has been performed and the structure has been determined to 3.0 Å resolution. The structure shows similarity to another proposed siderophore-interacting protein from S. putrefaciens and weak similarity to members of the NAD(P)H:flavin oxidoreductase superfamily.
The full length and the regulatory domain of the LysR-type transcriptional regulator CrgA have been crystallized. Diffraction data were collected from two crystal forms of full-length CrgA to 3.0 and 3.8 Å resolution, respectively. Crystals of the selenomethionine derivative of the C-terminal regulatory domain of CrgA diffracted to 2.3 Å resolution.
The crystal of the nsp3 ADRP domain of avian infectious bronchitis virus (IBV) has been obtained and subjected to further crystallograghic studies.
N-Acetylglucosamine 1-phosphate uridyltransferase (GlmU) from M. tuberculosis H37Rv has been crystallized and preliminary X-ray crystallographic analysis has been performed. GlmU is a bi-domained bifunctional enzyme that is involved in the biosynthesis of UDP-N-acetylglucosamine, a precursor in peptidoglycan biosynthesis in M. tuberculosis.
Par27 from B. pertussis, the prototype of a new group of parvulins has been crystallized in two different crystal forms.
G. zeae extracellular lipase has been overexpressed, purified and crystallized. Diffraction data were collected to 2.8 Å resolution.
The calcium-binding protein encoded by the CalD gene in S. coelicolor A3(2) has been crystallized as a prelude towards the determination of its three-dimensional structure by X-ray crystallography.
Crystallization and diffraction studies of human PCNA G178S mutant are reported.
The cloning, expression, purification and crystallization of the complex of the second and third regulatory subunits of human Pol δ are reported. The crystals were characterized and an X-ray diffraction data set was collected to a resolution of 3 Å.
Native and selenomethionine-derivatized CDCP2 from A. thaliana have been overexpressed, purified and crystallized. The crystals diffracted to 2.4 Å resolution.
The L-threonine dehydrogenase from T. kodakaraensis KOD1 has been crystallized in the tetragonal space group P43212, with unit-cell parameters a = b = 124.5, c = 271.1 Å. Diffraction data were collected to 2.6 Å resolution and preliminary analysis indicates that there are four molecules in the crystallographic asymmetric unit.
High-resolution diffraction was obtained from GluR4 AMPA-receptor ligand-binding domain crystals using a combination of cryogenic and room-temperature data-collection strategies.
The crystal structure of octopine dehydrogenase revealed a specific role of the His5 tag in inducing the crystal contacts required for successful crystallization.
A truncated variant of the human RuvBL1–RuvBL2 complex was cloned, expressed, purified and crystallised. Synchrotron diffraction data to 4 Å resolution were used to carry out a preliminary crystallographic analysis of the complex.
A hypothetical protein SCO4226 from S. coelicolor has been overexpressed, purified and crystallized. The crystals belonged to space group P21 and diffracted to 2.0 Å resolution.
Flavoredoxin from D. vulgaris Miyazaki F has been purified and crystallized. The crystals diffracted to 1.05 Å resolution using synchrotron radiation.
The X-ray structure of the guanidino kinase from S. mansoni has been determined at 2.8 Å resolution by the molecular-replacement method.
Succinate:ubiquinone oxidoreductase was solubilized and purified from E. coli inner membranes using several different detergents and the phospholipid content of each preparation was analyzed. The preparation with the lowest phospholipid content crystallized in two different crystal forms using detergent mixtures composed of n-alkyl-oligoethylene glycol monoether and n-alkyl-maltoside.
M. tuberculosis tetrahydrodipicolinate-N-succinyltransferase, the enzyme that catalyses the fifth reaction step of the lysine-biosynthesis pathway, has been cloned, expressed, purified and crystallized.
A crystal of the N-terminal domain of a plant NADPH oxidase was obtained and X-ray diffraction data were collected on a synchrotron beamline to a maximum resolution of 2.4 Å.
Recombinant RuBisCO chaperone from T. elongatus, TeRbcX, was aggregated and could not be crystallized. Mutations of an unusual Cys residue in the TeRbcX sequence yielded much better behaved proteins that could rapidly be crystallized.


 journal menu
journal menu











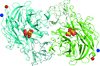























![[publBio]](/logos/publbio.gif)





