issue contents
June 2009 issue
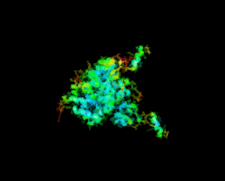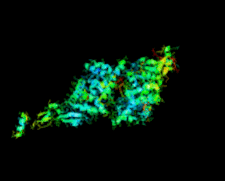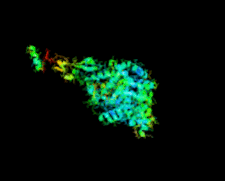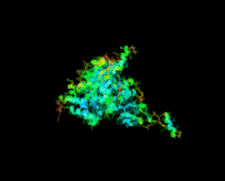
Cover illustration: Crystal structure of the GDNF-GFR![[alpha]](/logos/entities/alpha_rmgif.gif) 1 complex (Parkash & Goldman, p. 551).
1 complex (Parkash & Goldman, p. 551).
structural communications
The structure of AmpC β-lactamase (AmpCD) from E. coli with a tripeptide deletion (Gly286-Ser287-Asp288) is reported.
PDB reference: AmpCD, 2zj9, r2zj9sf
The crystal structure of an ATP-dependent DNA ligase from A. fulgidus has been determined. It resembles, but shows some variations from, the closed conformation of DNA ligase from P. furiosus.
PDB reference: A. fulgidus DNA ligase, 3gde, r3gdesf
The second crystal structure of the GDNF-GFRα1 complex provides further evidence that GFL signalling through RET is determined by the bend angle in the GFL.
PDB reference: GDNF–GFRα1 complex, 3fub, r3fubsf
crystallization communications
The PAS domain of RsbP, a stress-response protein from B. subtilis, was crystallized using the sitting-drop vapour-diffusion method. The crystals belonged to space group P21 and diffraction data were collected to a resolution of 1.6 Å.
NADPH-dependent L-sorbose reductase from G. frateurii (SR) was expressed, purified and crystallized with L-sorbose or NADPH using the sitting-drop vapour-diffusion method. Crystals of the SR–L-sorbose complex and SR–NADPH complex diffracted X-rays to 2.38 and 1.90 Å resolution, respectively.
RNA 3′-terminal phosphate cyclase from the crenarchaeon S. tokodaii has been overexpressed and purified. The apo form, its selenomethionine derivative and their complexes with ATP under different conditions have been crystallized. The structure of the selenomethionine-labelled form has been successfully solved by the Se-MAD method.
Vacuolar α-mannosidase, a cargo protein of the cytoplasm-to-vacuole targeting pathway, has been expressed, purified and crystallized.
The ribokinase Sa239 from S. aureus has been overexpressed in E. coli, purified and crystallized. Diffraction data were collected to 2.9 Å resolution.
The isolation, crystallization and preliminary X-ray crystallographic data of the transamidosome from T. thermophilus, a ribonucleoprotein particle that provides the translational machinery with Asn-tRNAAsn, are reported.
Oih-1, a β-lactamase enzyme isolated from the deep-sea bacterium O. iheyensis, has been crystallized and a complete X-ray diffraction data set has been collected to 1.65 Å resolution.
Full-length E. coli RNase G was overexpressed, purified and crystallized. Diffraction data were collected to a resolution of 3.4 Å.
Hibiscus chlorotic ringspot virus is a positive-sense monopartite single-stranded RNA virus that belongs to the Carmovirus genus of the Tombusviridae family. Authentic virus harvested from infected host kenaf leaves was purified and virus crystals were grown in multiple conditions. One of the crystals diffracted to 3.2 Å resolution and allowed the collection of a partial data set.
Interleukin-4-inducing principle from S. mansoni eggs (IPSE/alpha-1) was cloned, purified and crystallized using the hanging-drop vapour-diffusion method at 293 K. X-ray diffraction data were collected to 1.66 Å resolution.
Mouse RANKL and its receptor RANK have been cloned, expressed and purified. Crystals of RANK alone and in complex with RANKL have been obtained from which diffraction data have been collected to 2.0 and 2.8 Å resolution, respectively.
Uridine phosphorylase from S. typhimurium was expressed and purified and cocrystallized with the drug 5-fluorouracil. The crystals diffracted X-rays to 2.2 Å resolution using synchrotron radiation.
Two thioredoxins (SsTrxA2 and SsTrxA1) isolated from S. solfataricus have been crystallized. Diffraction data have been collected from SsTrxA2 and SsTrxA1 to 1.83 and 1.90 Å resolution, respectively. The structures of both enzymes have been solved by molecular replacement.
A secreted bacterial protein from a human pathogen that mediates serum resistance and adherence was overexpressed, purified, refolded and crystallized. Preliminary X-ray diffraction data are presented.
The ParE subunit of topoisomerase IV from X. oryzae pv. oryzae was expressed, purified and crystallized. Diffraction data were collected to a resolution of 2.15 Å.
Crystals of the V domain of human nectin-2 diffracted to 1.85 Å resolution and were monoclinic, belonging to space group P21, with unit-cell parameters a = 52.3, b = 43.9, c = 56.1 Å, β = 118.2°.
The expression, purification and crystallization of methionyl-tRNA synthetase from Mycobacterium smegmatis. The crystals diffracted to 2.1 Å.
The modified N-domain of the Wilson disease protein has been crystallized both in its native form and as a selenomethionine derivative. X-ray diffraction data were collected from the native crystal to 1.7 Å resolution, while the selenomethionine derivative diffracted to 2.7 Å resolution.
Conditions are reported for the crystallization of the flexible nuclear import receptor importin-β. Preliminary X-ray diffraction data indicate that the structure can be solved using molecular replacement.
Recombinant V. cholerae EpsG has been expressed, purified and crystallized. The crystals diffracted to 2.26 Å resolution.
β-L-Arabinopyranosidase from S. avermitilis NBRC14893 was crystallized by the sitting-drop vapour-diffusion method. The crystals diffracted to 1.6 Å resolution and belonged to space group P212121.
A novel 15 kDa antifungal protein amaryllin has been crystallized using 30% PEG 8000 as the precipitating agent. The crystals belong to orthorhombic space group I or I212121 with cell dimensions, a = 48.6, b = 61.9 and c = 79.6–Å.
Industrially used carbohydrate oxidase was successfully crystallized in several forms, diffraction data suitable for structural analysis were collected.
The gene coding for Q4DV70 has been cloned and the protein overexpressed in Escherichia coli with an N-terminal His tag. Purification of Q4DV70 was carried out by affinity and size-exclusion chromatography and the His tag was removed by TEV protease digestion. Crystals of Q4DV70 were grown using the sitting-drop vapour-diffusion method.
The NADPH-dependent 3-quinuclidinone reductase from Rhodotorula rubra was expressed, purified, and crystallized and X-ray diffraction data of this crystal were collected to 2.2 Å resolution.
RIG-I is important in recognizing viral RNA and stimulating the immune system. Human RIG-I protein was crystallized in complex with 18-mer double-stranded RNA to study the recognition mechanism.
Grx5 from Saccharomyces cerevisiae is a monothiol glutaredoxin that is involved in iron–sulfur cluster biogenesis. Here, yeast Grx5 was cloned and overproduced in Escherichia coli and the purified protein was crystallized using the hanging-drop vapour-diffusion method.


 journal menu
journal menu











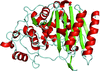


























![[publBio]](/logos/publbio.gif)





