issue contents
November 2010 issue
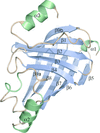
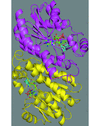
Cover illustration: Structure of Plasmodium falciparum ADP-ribosylation factor 1, p. 1426.
editorial
Free 

structural communications
The crystal structure of phenolic acid decarboxylase from B. pumilus strain UI-670 has been determined and refined at 1.69 Å resolution. The enzyme is a dimer, with each subunit adopting a β-barrel structure belonging to the lipocalin fold.
PDB reference: phenolic acid decarboxylase, 3nad
The X-ray structure of human bisphosphoglycerate mutase was solved to 1.94 Å resolution and a bisphosphoglycerate mutase–citrate model was built to explore enzyme–inhibitor interactions.
PDB reference: bisphosphoglycerate mutase, 3nfy
The homodimeric nature of the ESAT-6 homologue GBS1074 and the potential for fibre-like assemblies are revealed by the 2 Å resolution crystal structure.
PDB reference: homodimeric GBS1074, 3o9o
The crystal structure of GDP-bound ARF1 GTPase of Plasmodium falciparum has been determined at 2.5 Å resolution and compared with the structures of mammalian ARF1s.
PDB reference: ARF1 GTPase, 3lrp
A comparison is made between the structures of dihydroorotase from four different organisms, including B. anthracis dihydroorotase, and reveals substantial variations in the active site, dimer interface and overall tertiary structure.
PDB reference: dihydroorotase, 3mpg
Structure of the ternary complex of F. tularensis enoyl-acyl carrier protein reductase reveals the structure of the substrate binding loop whose electron density was missing in an earlier structure, and demonstrates a shift in the position of the NAD+ cofactor.
PDB reference: enoyl-acyl carrier protein reductase, 3nrc
crystallization communications
The invertase from X. dendrorhous has been purified, deglycosylated and crystallized and diffraction data have been collected to 2.3 Å resolution.
Recombinant nucleoside triphosphate hydrolases from N. caninum and T. gondii have been purified and crystallized for X-ray structure analysis.
A marine diatom-infecting virus was crystallized using the hanging-drop vapour-diffusion method. The crystals belonged to space group P6322, with unit-cell parameters a = b = 448.67, c = 309.76 Å, and diffracted to 4.0 Å resolution.
Cleavage of the serpin IRS-2 from the hard tick I. ricinus by contaminating proteolytic activity mimicked the specific processing of the serpin by its target protease and resulted in a more stable form of the serpin which produced crystals that diffracted to 1.8 Å resolution.
Virions of red clover necrotic mosaic virus have been purified and crystallized. The space group was determined to be I23, with unit-cell parameter a = 377.8 Å. The crystals diffracted to 4 Å resolution.
The expression, purification, crystallization and preliminary X-ray diffraction analysis of CTP:inositol-1-phosphate cytidylyltransferase from A. fulgidus is described.
The crystallization and preliminary diffraction analysis of LMO2 in complex with the LID domain of Ldb1 is described. A three-wavelength anomalous dispersion (MAD) data set was collected to 2.8 Å resolution at the Zn K edge.
Dissimilatory sulfite reductase from Desulfovibrio vulgaris Miyazaki F was purified and crystallized. The crystals belonged to the space group P41212 and diffracted to 3.7 Å resolution.
PA3885 (TpbA), a tyrosine phosphatase, may function as a balancing factor between biofilm formation and motility in the opportunistic pathogen P. aeruginosa. Here, the expression, purification, crystallization and preliminary crystallographic analysis of PA3885 from P. aeruginosa PAO1 are reported.
Dimeric and trimeric cytochromes c were purified and crystallized. X-ray diffraction data sets were collected to resolutions of 2.2 and 2.1 Å, respectively.
The terminal oxygenase component (Oxy) of carbazole 1,9a-dioxygenase (CARDO) catalyzes dihydroxylation of the aromatic ring. The Oxy of CARDO from Novosphingobium sp. KA1 was crystallized and the crystals diffracted to a resolution of 2.1 Å.
RlmL, a 23S rRNA m2G2445 methyltransferase from Escherichia coli, was expressed, purified and crystallized, and crystals diffracted to 2.2 Å.
The expression, purification, crystallization and preliminary X-ray diffraction analysis of the soluble domain of human glioma pathogenesis-related protein 1 (sGLIPR1) is presented.
In this study, human NDPK-A has been crystallized under oxidative conditions and X-ray data have been collected to 2.80 Å resolution using synchrotron radiation.
TrpY was crystallized using the hanging-drop method with ammonium sulfate as the precipitant. The crystals belonged to the tetragonal space group P43212 or P41212, with unit-cell parameters a = b = 87, c = 147 Å, and diffracted to 2.9 Å resolution.
The E. coli TehB methyltransferase has been purified and crystallized in the presence of SAM and sinefungin. Diffraction data have been collected to 1.9 Å resolution for both complexes.
3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase of Streptococcus pneumoniae has been cloned, overexpressed and purified to homogeneity using Ni–NTA affinity chromatography. Crystals were obtained using the hanging-drop vapour-diffusion method.
The carbohydrate-binding region of GspB from S. gordonii strain M99 was crystallized in space group P212121 and data were collected to 1.3 Å resolution.
A novel dye-linked L-proline dehydrogenase from a hyperthermophilic archaeon was successfully isolated and crystallized.
Dihydrodipicolinate synthase (DHDPS) is an essential oligomeric enzyme of interest to antibiotic discovery research and studies probing the importance of quaternary structure to protein function, stability and dynamics. The cloning, expression, purification and crystallization of DHDPS from the psychrophilic (cold-dwelling) bacterium Shewanella benthica are described.
A 29 kDa cis-biphenyl-2,3-dihydrodiol-2,3-dehydrogenase from P. pnomenusa B-356 was purified, crystallized and characterized by the X-ray diffraction method.
The crystallization and preliminary X-ray analysis of recombinant human cytosolic seryl-tRNA synthetase (hsSerRS) is reported. Diffraction data were collected to 3.13 Å resolution and the hsSerRS crystal structure is currently being refined and analyzed.
The outer membrane channel protein Rv1698 from M. tuberculosis has been overexpressed in E. coli, purified and crystallized. Diffraction data were collected to 2.5 Å resolution.
The crystallization and diffraction studies of EcoO109I DNA methyltransferase are described.
Achromobacter protease I (API), a lysine-specific serine protease, exhibits an order of magnitude higher activity than bovine trypsin, while its optimum pH is in the alkaline region at approximately pH 9. Two single point mutants have previously been reported to improve the optimum pH range; determination of the tertiary structure of API mutants would help in understanding these mechanisms.
The SicA protein from the motile enterobacterium S. enterica serovar Typhimurium was overexpressed and crystallized. The crystals diffracted to 3.5 Å resolution and belonged to the monoclinic space group C2, with unit-cell parameters a = 180.4, b = 94.1, c = 131.8 Å, β = 130.9°. The crystallographic asymmetric unit may contain as many as eight molecules, suggesting a unique oligomerization state.
The major haemoglobin of the sub-Antarctic fish E. maclovinus is the first sub-Antarctic fish haemoglobin to be crystallized and its structural characterization will shed light on the molecular mechanisms of cold adaptation and the role of the Root effect in fish.
The physiologically active form of nitrous oxide reductase was isolated and crystallized under strict exclusion of dioxygen and diffraction data were collected from crystals belonging to two different space groups.
Open  access
access
 access
accessThe crystallization of the recombinant protease from Southampton norovirus is described. Whilst the native crystals were found to diffract only to medium resolution (2.9 Å), cocrystals with a designed covalently bound inhibitor diffracted X-rays to 1.7 Å resolution.
addenda and errata
Free 



 journal menu
journal menu








































![[publBio]](/logos/publbio.gif)






