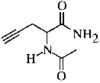
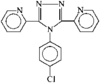
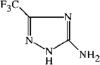
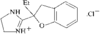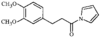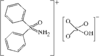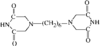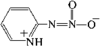issue contents
March 1998 issue
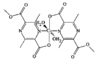
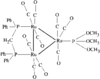
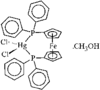
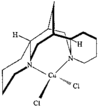
Cover illustration: A Novel Cyclotriphosphazene Complex with a Short Transannular P...P Contact, see Thomas, pages 331-333. Displacement ellipsoids are shown at the 50% probability level for non-H atoms and H atoms have been omitted for clarity.
cif-access (organic compounds)
Download citation


Download citation


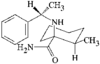
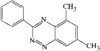
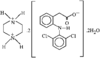
In the title compound (efaroxan hydrochloride), homochiral cationic molecules form chains along the b axis of the triclinic cell via N-H⋯Cl hydrogen bonds. The furanyl groups are nearly parallel to the a axis and the imidazolinium rings are roughly perpendicular to it.
Download citation


Download citation


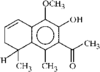
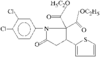
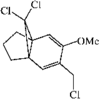
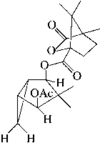
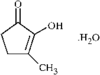
The structure and relative stereochemistry of O-methyldehydronorcacalol, a sesquiterpenoid isolated from Traversia baccharoides Hook f., is established. The hydroxyl group takes part in a bifurcated hydrogen bond, intramolecular with an adjacent methoxy O atom and intermolecular with an acetyl O atom of an adjacent molecule.
Download citation


Download citation


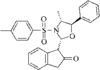
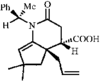
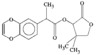
1,3-Oxazolidines are powerful chiral templates for auxiliary-directed asymmetric synthesis. The stereochemical parameters of the title compound are in good agreement with other arylsulfonyloxazolidines.
Download citation


Download citation


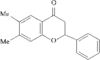
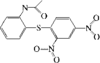
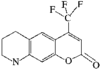
In the title molecule, the dihydropyran ring adopts a sofa conformation. The benzopyran ring is distorted from planarity and the phenyl ring forms a dihedral angle of 53.86 (6)° with the dihydropyran ring.
inorganic compounds
Download citation


Download citation


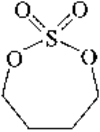
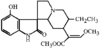
A new synthetic route for the preparation of single crystals of monoclinic VOHPO4.2H2O has been found. The results of the single-crystal structure analysis are compared with those of an earlier X-ray powder structure determination.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu