issue contents
April 2000 issue
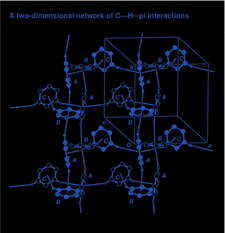
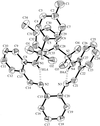
Cover illustration: The bilayer of C-H
![[pi]](/logos/entities/pi_rmgif.gif) -bonded aromatic rings in 3-phenyl-1-(1-phenylethyl)-4-(1-phenylethyliminomethyl)azetidin-2-one. Only the aromatic C atoms and H atoms involved in the interactions are shown. See Kooijman et al. [Acta Cryst. C56, 481-483].
-bonded aromatic rings in 3-phenyl-1-(1-phenylethyl)-4-(1-phenylethyliminomethyl)azetidin-2-one. Only the aromatic C atoms and H atoms involved in the interactions are shown. See Kooijman et al. [Acta Cryst. C56, 481-483].
inorganic compounds
Download citation


Download citation


The structure of HgWO4 consists of zigzag chains of edge-sharing WO6 and HgO6 octahedra running parallel to the c axis. Hg characteristically forms two short collinear Hg—O bonds.
Download citation


Download citation


The title compound contains iron(II) octahedrally coordinated by diphosphates and water molecules. The octahedra are connected in such a way that they form channels throughout the structure which contain the coordinated water molecules.
Download citation


Download citation


Rietveld refinements of monoclinic and triclinic AlPO4 tridymites show that their tetrahedral framework structures are isotypic with the silica analogues. The two forms of tridymite differ in distortions of the six-membered rings of tetrahedra, resulting in different superstructures. Both modifications co-exist at room temperature.
Download citation


Download citation


SrCrP2O7 belongs to the structure family of α-Ca2P2O7. It consists of a three-dimensional network of [SrO9], [CrO5] and [P2O7] groups. Lattice constants of the whole series of isotypic compounds SrMP2O7 (M2+ = Cr, Zn or Cd) are compared with respect to the electronic configuration of M2+.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Free 

electronic papers (inorganic compounds)
Download citation


Download citation


Nonapotassium cerium tetraphosphorus hexadecasulfide is a zero-dimensional material isostructural with the rubidium analogue.
electronic papers (metal-organic compounds)
Download citation


Download citation


Recrystallization of trans-[Co(acac)2(PMePh2)(H2O)]PF6 (acac is pentane-2,4-dionato) from aqueous methanol gave purple prisms of the title compound. The molecular structure of the complex cation having a coordinated methanol ligand was revealed with a Co—O(methanol) distance of 2.059 (3) Å.
Download citation


Download citation


The title Nd compound is isomorphous with the corresponding Y compound, but with a larger unit cell.
Download citation


Download citation


The coordination geometry around the Ni atom is square planar with a slight tetrahedral distortion. The five-membered chelate ring has a distorted gauche conformation with the phenyl groups in axial and equatorial orientations.
Download citation


Download citation


The title complex shows a distorted octahedral coordination and has a crystallographic twofold rotation axis. Intermolecular O—H⋯O hydrogen bonds form a two-dimensional network in the ab plane.
Download citation


Download citation


The title compound, (Et4N)[ZnI4], is isostructural with the previously reported tetrabromocadmate, whose coordinates were used as the starting parameters for both of the refinements presented here. The asymmetric unit comprises one anion and three cations, all of which are in special positions. The anion has five nearest-neighbour cations in the form of a square pyramid.
Download citation


Download citation


Monoclinic and triclinic forms of bis[2-hydroxy-2,2-bis(2-pyridyl)ethanoato-O1,N,N′]nickel(II)
Both the monoclinic and triclinic forms of the neutral title complex show an octahedral nickel(II) centre coordinated by two facial tridentate pyridylate ligands. The O-donor atoms of each ligand are trans.
Download citation


Download citation


The CoIII ion is six-coordinate in a nearly octahedral environment (one Cl, one O and four N atoms) and the ZnII ion is four-coordinate in a distorted tetrahedral environment (three Cl and one O atom).
Download citation


Download citation


The central Co atom of the title complex has a distorted octahedral coordination. The three Co—O bond distances are in the range 1.887 (2)–1.910 (2) Å, while the three Co—N bond distances are in the range 1.919 (2)–1.934 (2) Å. The ethanol solvate molecule is involved in an O—H⋯O intermolecular hydrogen bond.
Download citation


Download citation


The crystal structure of the title compound contains discrete monomeric molecules of the carbene complex and solvent molecules separated by normal van der Waals distances. The Pd atom is four-coordinate in an essentially square-planar environment, with the chlorine ligands mutually cis; Pd—P = 2.2495 (7), Pd—Cl = 2.3508 (7) and 2.3600 (7), Pd—C = 1.948 (2) and N—C(carbene) = 1.274 (3) Å.
Download citation


Download citation


The title compound crystallizes as discrete ions, which form interionic hydrogen bonds of the type N—H⋯F.
electronic papers (organic compounds)
Download citation


Download citation


In the title, compound, both chiral centers have the an S configuration. Both OH groups and both H atoms of the water molecule form intermolecular hydrogen bonds with O⋯O distances in the range 2.7686 (18)–2.8717 (18) Å.
Download citation


Download citation


The absolute configuration at the new stereogenic centre, formed during the key step of the total synthesis, was established by X-ray analysis of the title compound.
Download citation


Download citation


In crystals of the title compound, the hydrogen fumarate anions form one-dimensional chains through O—H⋯O hydrogen bonding along the c and (a+b)/2 directions. There are three sites of the hydrogen fumarate, two of which have an inversion centre.
Download citation


Download citation


The absolute configuration was determined for the title compound, which was prepared in a synthetic study on the natural products bryostatins. There are two independent molecules which show similar conformations, except for the orientation of the methoxy groups.
Download citation


Download citation


The absolute configuration was determined for the optically active title compound, which was prepared in a synthetic study on the natural products bryostatins.
Download citation


Download citation


In the four title compounds, (I)–(IV>), respectively, the donor and acceptor molecules are stacked alternately to form one-dimensional columns, but with different relative orientations of the alkyl substituents and thenitro groups.
Download citation


Download citation


In the two title compounds, (I>) and (II>), respectively, the donor and acceptor molecules are stacked alternately to form one-dimensional columns, but with different relative orientations of the substituents on the donor andacceptor.
Download citation


Download citation


The three title structures, (I)–(III>), respectively, consist of 4-(dimethylamino)pyridinium and dinitrobenzoate ions connected by an N—H⋯O hydrogen bond. Donor–acceptor π–π stacking is observed in (II>) and (III>), but notin (I>).
Download citation


Download citation


The title amide does not photocyclize in the solid state. The methoxy group is involved in intermolecular steric interactions and so prevents rotation of the N-phenyl group in the crystal.
Download citation


Download citation


The crystal structures of the two title thiobenzamides, i.e. (I>) and (II>), were determined to investigate the relationship between the photoreactivity in solid state and the structure. Their geometry was confirmed to be the E isomer in each case.
Download citation


Download citation


The crystal structure of the title thiobenzamide was determined to investigate the relationship between the photoreactivity in solid state and the structure. The geometry was confirmed to be the Z isomer.
Download citation


Download citation


The title compound is the photoproduct obtained from N-isopropyl-N-[(E)-2-phenylpropenyl]thiobenzamide. Recrystallization showed a spontaneous resolution.
Download citation


Download citation


The title compound is a double mesogen in which two rigid p-terphenyl-based units are connected laterally by a carbonate linker. Layers of molecules pack along the a axis, stimulating a smectic A-type arrangement, and on melting, a smectic A phase appears at 388 K and becomes isotropic at 389 K.
Download citation


Download citation


N-3,5-Dinitrobenzoyl-L-leucine crystallizes as two independent molecules which differ in their conformation. Intermolecular hydrogen bonding involving (amide)N—H⋯O=C(acid) and (acid)O—H⋯O=C(amide) interactions results in a two-dimensional network consisting of hydrogen-bonded rings with graph-set R44(20).
Download citation


Download citation


A new polymorphic form of N,N′-diphenyl-1,4-phenylenediamine has been characterized. There are two independent centrosymmetric molecules, both of which are more non-planar than the orthorhombic form.
Download citation


Download citation


A high degree of bending of the two five-membered rings in the title compound produces close contacts between two C atoms in each of the benzo groups and one S atom in the other dithiole ring.
Download citation


Download citation


The title compound crystallizes with two molecules in the asymmetricunit, which are held together by an extended network of hydrogen bonds.Only five of the six theoretically possible hydrogen bonds areformed.
Download citation


Download citation


The 11-membered ring of the title compound, which may be used for complexing metal atoms, adopts a chair conformation of approximate mirror symmetry.
Download citation


Download citation


The diazepine ring of the title compound adopts a boat conformation. An intramolecular hydrogen bond is formed between the NH group of the diazepine ring and a carbonyl O atom of one of the side chains.
Download citation


Download citation


The title compound is being used as a platform for new tripodal receptors.Two molecules make up the asymmetric unit and weak intermolecularhydrogen bonding is observed between a methylene H atom and theCl atom of an adjacent molecule; there are also C—H⋯π interactions.
Download citation


Download citation


The two bicyclic fragments of the molecule are planar and are turned with respect to each other by 77.8 (2)°. The formation of intramolecular O—H⋯O and N—H⋯O hydrogen bonds causes considerable changes in the bond lengths within the amidopyridine fragment.


 journal menu
journal menu