issue contents
January 2013 issue
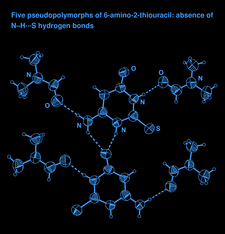
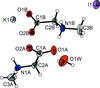
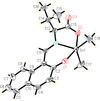
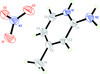
Cover illustration: A perspective view of 6-amino-2-thiouracil dimethylformamide disolvate, showing the atom-numbering scheme. Displacement ellipsoids are drawn at the 50% probability level and H atoms are shown as small spheres of arbitrary radii. The dashed lines indicate N-H O hydrogen bonds. See Hützler & Bolte [Acta Cryst. (2013), C69, 93-100].
O hydrogen bonds. See Hützler & Bolte [Acta Cryst. (2013), C69, 93-100].
inorganic compounds
Download citation


Download citation


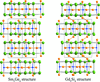
Three members of the RE5−xLixGe4 (x ≃ 1) family have been structurally characterized. Their crystal chemistry mirrors that of RE5−xMgxGe4 (x ≃ 2), and the valence electron count can be understood on the basis of the Zintl–Klemm formalism.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu