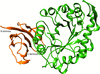issue contents
November 2016 issue

Cover illustration: The NIH BD2K Integrated Resource for Reproducibility in Macromolecular Crystallography (IRRMC) is a freely accessible data repository for diffraction data (Grabowski et al., p. 1181). It currently hosts over 3000 diffraction experiments and associated metadata. The IRRMC was created for preserving original diffraction images in order to increase the reproducibility, transparency and scientific integrity of crystallographic experiments. IRRMC utilizes a federated architecture of many independent storage servers, which provides both scalability and sustainability.
research papers
The thermostable glucuronoxylan endo-β-1,4-xylanase from C. thermocellum cleaves the xylan chain specifically at sites containing 4-O-methylglucuronic acid substitutions. The structure of the ligand-bound enzyme mutant E225A solved at 1.17 Å resolution shows binding of the xylotetraose-cleavage oligosaccharides at subsites −3 to +2.
Open  access
access
 access
accessThe first crystal structure of ThcOx, a cyanobactin oxidase, has been determined to 2.65 Å resolution using S-SAD phasing. This is the first structure reported from the purpose-designed long-wavelength beamline I23 at Diamond Light Source.
PDB reference: ThcOx, 5lq4
The Integrated Resource for Reproducibility in Macromolecular Crystallography (IRRMC) is a large, scalable, and searchable web-accessible archive of protein crystallography diffraction experiments organized according to metadata.
Open  access
access
 access
accessPerdeuteration, purification and the growth of large crystals of the carbohydrate-recognition domain of galectin-3C are described. Five neutron diffraction data sets have been collected at four neutron sources; these are compared and two are merged.
The crystal structure of racemic Z-DNA solved at an ultrahigh resolution of 0.78 Å in a centrosymmetric space group shows the massive presence of multiple conformations, not only of the backbone but also at some base pairs. The structure also reveals novel hydration patterns and an unusual double-helix packing pattern.
PDB reference: centrosymmetric Z-DNA, 5jzq
Open  access
access
 access
accessThe crystal structure of the liver isoform of human fructose-1,6-bisphosphatase in the active R-state conformation was determined by molecular replacement using data from a crystal with noncrystallographic rotational symmetry and pseudo-translation. Owing to an almost perfect placement of noncrystallographic symmetry elements, quadruple space-group ambiguity within the same Laue symmetry arises, including two enantiogenic pairs. The origins of space-group ambiguity, the assignment of the correct space group, refinement and model properties are discussed.
PDB reference: human liver FBPase, 5ldz
book reviews
Free 



 journal menu
journal menu