issue contents
September 2009 issue
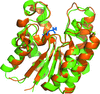
Cover illustration: Escherichia coli malate dehydrogenase at 1.45 Å resolution (Zaitseva, Meneely & Lamb, p. 866).
structural communications
Here, the crystal structure of adenylosuccinate lyase from Escherichia coli was determined to 1.9 Å resolution.
PDB reference: PurB, 3gzh, r3gzhsf
The crystal structure of the esterase EstA from a cold-adapted bacterium was determined in a form that was covalently inhibited by monoethylphosphonate.
PDB reference: EstA, 3hp4, r3hp4sf
The structure of apo malate dehydrogenase from Escherichia coli has been determined to 1.45 Å resolution.
PDB reference: malate dehydrogenase, 3hhp, r3hhpsf
crystallization communications
Crystals of the N-terminal domain of Gram-negative bacteria-binding protein 3 of D. melanogaster grown from PEG solutions are monoclinic (space group C2) and diffract to 1.7 Å resolution.
The βC-S lyases from two oral bacteria, Streptococcus anginosus and S. gordonii, were cloned, overproduced, purified and crystallized. The obtained crystals were characterized by X-ray diffraction.
The crystallization of the metallo-cofactor (R2F) of native ribonucleotide reductase isolated from the Mn-requiring Gram-positive bacterium C. ammoniagenes is described. The crystals diffracted to 1.36 Å resolution.
An all-LNA duplex was designed from the stem region of an RNA aptamer which has been generated against ricin. The LNA duplex was crystallized and preliminary X-ray diffraction analysis revealed diffraction to a resolution of up to 2.8 Å.
Recombinant 4-pyridoxolactonase from M. loti MAFF303099 was crystallized in two forms and diffraction data were collected to 2.0 and 1.9 Å resolution, respectively.
To understand the role of the Cdc7–Dbf4 complex in checkpoint responses, a fragment of Saccharomyces cerevisiae Dbf4 encompassing motif N was isolated, overproduced and crystallized.
Galactose-binding lectin from champedak was crystallized at 293 K. Preliminary X-ray diffraction analyses are reported.
Open  access
access
 access
accessThe crystallization of a novel controller protein is reported and its interaction with DNA is characterized.
The crystallization and preliminary X-ray diffraction studies of an endo-1,5-α-arabinanase from hyperthermophilic T. petrophila are reported. The crystals diffracted to 2.86 Å resolution.
The proliferating cell nuclear antigen (PCNA) from a novel hyperthermophilic archaeon Thermococcus thioreducens has been crystallized, and diffraction data have been collected to 1.86 Å.
Single orthorhombic crystals of M. genitalium protein MG289 have been grown and shown to diffract X-rays to 2.8 Å resolution with good statistics. The structure obtained from these data will help to provide insight into the function of the protein as well as improving the understanding of its role in this human pathogen.
Poly-γ-glutamate hydrolase from bacteriophage ΦNIT1 was crystallized by the sitting-drop vapour-diffusion method and the crystals diffracted to beyond 2.4 Å resolution.
A new alkaline phosphatase enzyme from Sphingomonas sp. strain BSAR-1, termed PhoK, has been shown to be useful in uranium bioprecipitation. PhoK has been expressed, purified and crystallized.
Crystals of the soluble form of the [NiFeSe] hydrogenase from D. vulgaris Hildenborough were obtained and belonged to the monoclinic space group P21, with unit-cell parameters a = 60.57, b = 91.05, c = 66.85 Å, β = 101.46°.
Flap endonuclease 1 from D. amylolyticus was expressed, purified and crystallized by the sitting-drop vapour-diffusion method. X-ray diffraction data were collected to 2.00 Å resolution.
Adenylate kinase (AK) from D. gigas was purified and crystallized in three different metal-bound forms: Zn2+–AK, Co2+–AK and Fe2+–AK.
The ubiquitin-like domain of human hHR23A protein was crystallized by the hanging-drop vapour-diffusion method in space group P6522 and diffraction data were collected to 1.97 Å resolution. Structure solution by molecular replacement is described.
Aspartate transcarbamoylase, the second enzyme of the de novo pyrimidine-biosynthetic pathway, from T. cruzi has been purified and crystallized for X-ray structure analysis.
The cloning, overexpression, purification, crystallization and preliminary X-ray crystallographic analysis of glyceraldehyde-3-phosphate dehydrogenase from A. mylitta are reported.
Rhodoquinol-fumarate reductase is a key enzyme in the anaerobic respiratory chain of adult A. suum mitochondria. Its crystallization in the presence of a mixture of octaethyleneglycol monododecyl ether and n-dodecyl-β-D-maltopyranoside in a form suitable for X-ray structure analysis is reported.
Complexes of both hIGF-II and hIGF-IIE with a Fab have been crystallized and investigated by X-ray analysis.
Jack bean urease was purified and crystallized and X-ray diffraction data were collected to 2.05 Å resolution.
Leucine aminopeptidase, an exopeptidase that hydrolyzes leucine from the N-terminus of polypeptides, from X. oryzae pv. oryzae was cloned, expressed and crystallized.
The cloning, purification and crystallization of the C-terminal domain of human hPrp22 are reported. This communication also contains data for the preliminary X-ray diffraction analysis.


 journal menu
journal menu





































![[publBio]](/logos/publbio.gif)





