issue contents
June 2015 issue
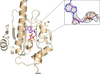
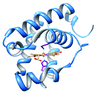
Cover illustration: N-terminal domain of heat-shock protein 90 from Oryza sativa (Raman & Suguna, p. 688).
IYCr crystallization series
Open  access
access
 access
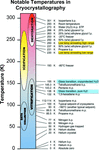
accessCurrent methods, reagents and experimental hardware for successfully and reproducibly flash-cooling macromolecular crystals to cryogenic temperatures for X-ray diffraction data collection are reviewed.
research communications
Hookworm platelet inhibitor belongs to the cysteine-rich/antigen-5/pathogenesis-related 1 (CAP) protein family. It has been linked to integrin-antagonist, antiplatelet activity found in extracts of A. caninum hookworms.
PDB reference: hookworm platelet inhibitor, 4tpv
The crystal structure of the catalytic domain of human dual-specificity phosphatase 7 (C232S) is presented at 1.67 Å resolution.
The crystal packing of N,N′-diacetylchitobiose deacetylase from P. furiosus varied depending on the presence of cadmium and whether the native protein or its selenomethionine derivative was used. The cadmium complex was involved in the crystal contacts in Cd-containing conditions.
The CFA/III minor pilin subunit, CofB, has been crystallized. An N-terminal hydrophobic portion of CofB was truncated to enable initial crystallization. Dehydration of preliminary crystals yielded higher resolution diffraction patterns.
Crystallization and diffraction studies of the HIRAN domain of human HLTF are reported.
The lantibiotic-resistance protein NSR from S. agalactiae was crystallized in two different crystal forms, which diffracted X-rays to a maximum resolution of 2.2 Å.
Open  access
access
 access
accessIn Escherichia coli, the common pilus (Ecp) belongs to an alternative chaperone–usher pathway that plays a major role in both early biofilm formation and host-cell adhesion. Initial attempts at crystallizing the chaperone EcpB using natively purified protein from the bacterial periplasm were not successful; however, after the isolation of EcpB under denaturing conditions and subsequent refolding, crystals were obtained at pH 8.0 using the sitting-drop method of vapour diffusion. This is the first time that this refolding strategy has been used to purify CU chaperones.
A β-1,3-glucanase from the thermophilic fungus C. thermophilum was overexpressed in P. pastoris, purified and crystallized. In situ diffraction at a synchrotron and seeding were used to obtain reproducible crystals. A complete data set to 2.0 Å resolution was collected using an in-house X-ray generator.
The crystal structure of the LRR domain of PP32A has been solved to 1.6 Å resolution.
PDB reference: human PP32A LRR domain, 4xos
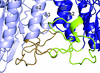
Binding studies and an activity assay were performed for heat-shock protein 90 from O. sativa. The N-terminal domain of Hsp90 was crystallized and its structure was determined in complex with AMPPCP.
PDB reference: heat-shock protein 90, 4x9l
Crystals of a complex between human B7-H6, a tumor cell ligand for NKp30, and an inhibitory monoclonal antibody have been obtained. The crystals diffracted to 2.5 Å resolution.
Contact-dependent growth inhibition (CDI) is an important mechanism of intercellular competition between Gram-negative bacteria. The X-ray crystal structure of an immunity protein from N. meningitidis MC58 is presented together with a structural model of the toxin–immunity protein complex.
PDB reference: CdiI immunity protein, 4q7o
Crystallization of apo interleukin-18 in low ionic strength conditions was achieved through surface-entropy reduction and rational protein design. Here, the design, expression, purification, crystallization, and structure determination of three apo interleukin-18 structures to between 1.9 and 2.85 Å resolution are reported.
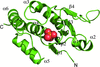
The structure of THB1, a monomeric 2/2 hemoglobin involved in nitrogen metabolism in C. reinhardtii, was solved to 1.9 Å resolution. The heme is coordinated by the proximal histidine and a distal lysine.
PDB reference: Chlamydomonas reinhardtii THB1, 4xdi
The X-ray structure of cyanide-bound bovine heart cytochrome c oxidase in the fully oxidized state was determined at 2.0 Å resolution. This structure shows that a cyanide ion bridges the two metals in a nearly symmetric end-on fashion.
PDB reference: bovine heart cytochrome c oxidase, 3x2q
A MepR-like transcription factor, BC0657, from B. cereus was expressed, purified and crystallized. X-ray diffraction data for the BC0657 crystals were collected to a resolution of 2.05 Å.
A Cmr1-deficient functional Cmr complex composed of P. furiosus Cmr2–Cmr3, A. fulgidus Cmr4–Cmr5–Cmr6 and the 39-mer P. furiosus 7.01-crRNA was cocrystallized with single-stranded DNA complementary to the crRNA guide. X-ray diffraction data for the crystals were collected to 2.1 Å resolution using a synchrotron-radiation source.
The crystal structure of the M. tuberculosis MarR family protein Rv0880 is described.
PDB reference: Rv0880, 4yif
Open  access
access
 access
accessLatent and active aurone synthase purified from petals of C. grandiflora (cgAUS1) were crystallized. The crystal quality of recombinantly expressed latent cgAUS1 was significantly improved by co-crystallization with the polyoxotungstate Na6[TeW6O24] within the liquid–liquid phase-separation zone.
The crystal structure of the Mre11 nuclease dimer from the thermophilic eukaryote C. thermophilum is reported. Crystals of C. thermophilum Mre11, which plays an important role in DNA double-strand break repair, diffracted to 2.8 Å resolution and the structure was solved by the molecular-replacement technique.
PDB reference: Mre11 catalytic domain, 4yke
3-Ketoacyl-CoA thiolase from R. eutropha (ReH16_A1887), an enzyme involved in β-oxidation degradative pathways, has been crystallized and X-ray diffraction data have been collected from the crystal and analyzed.
Nicotinamide adenine dinucleotide synthetase (NAD synthetase) from methicillin-resistant S. aureus was cloned, purified and crystallized, and X-ray diffraction data were collected to a resolution of 2.0 Å.
The A. baumannii biofilm-forming Csu fimbriae are assembled via the archaic CsuC–CsuD chaperone–usher pathway. To determine the structural basis for the biogenesis of Csu fimbriae, native and selenomethionine-substituted CsuC–CsuA/B chaperone–major subunit pre-assembly complexes were produced in Escherichia coli, purified and crystallized. Initial phases were derived from a single anomalous diffraction (SAD) experiment using the selenomethionine derivative.
X-ray data for eight different Dscam1 Ig1–4 isoforms were collected to 1.9–4.0 Å resolution. These structures will provide the opportunity to perform extensive structural comparisons of different Dscam1 isoforms and will provide insight into its specificity.
The purification, crystallization and molecular-replacement structure solution of two crystal forms of the major acid phosphatase from L. pneumophila are described.
X-ray data for the R. flavefaciens cell surface carbohydrate-binding protein CttA, which is involved in the binding of the whole bacterium to cellulose, have been collected to resolutions of 3.23 and 1.61 Å in trigonal and monoclinic space groups, respectively. The structure was phased using bound iodide from the crystallization buffer.
In order to understand the structure–function relationship of 3-deoxy-D-manno-oct-2-ulosonic acid 8-phosphate phosphatase (YrbI) from B. pseudomallei, its crystallographic study has been performed. Here, the cloning, overexpression, purification, crystallization and preliminary X-ray study of this enzyme are reported.
Diffraction data from crystals of zebrafish (D. rerio) β2-microglobulin type I and type II (Dare-β2m-I and Dare-β2m-II) were collected to 1.6 and 1.9 Å resolution, respectively.
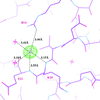
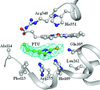
The crystal structures of three ternary complexes of human (h) or P. carinii (pc) dihydrofolate reductase (DHFR) with cofactor and pyrido[2,3-d]pyrimidine inhibitors are reported to define the interactions that enhance selectivity against pathogenic DHFRs. The binding conformation of the most potent inhibitor differs between the human and Pneumocystis complexes and validates the hypothesis that the N9-methyl substitution preferentially interacts with Ile123 of pcDHFR than with Val115 of hDHFR, consistent with its weaker binding affinity for hDHFR.
addenda and errata
Free 

The article by Singh et al. [(2015), Acta Cryst. F71, 304–310] is retracted.


 journal menu
journal menu











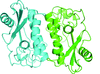

















![[publBio]](/logos/publbio.gif)





