issue contents
May 2016 issue
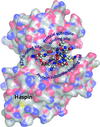
Cover illustration: RRM domain of human RBM7 (Sofos et al., p. 397). In eukaryotic cells, RNA surveillance, maturation and decay depend to a great extent on the 3'-5' exonucleolytic and endonucleolytic activities of the RNA exosome complex. The RRM-protein RBM7 is part of the nuclear exosome targeting (NEXT) complex that directs the nuclear RNA exosome to many of its nucleoplasmic substrates.


 journal menu
journal menu
















![[publBio]](/logos/publbio.gif)





