issue contents
March 2017 issue
Cover illustration: AHpmA265 (truncated hemolysin A) from Proteus mirabilis (Novak et al., p. 138).
research communications
Open  access
access
 access
accessThe recent epidemic of Zika virus (ZIKV) has created a major global health concern. Here, the crystal structure of the full-length ZIKV NS5 protein at 3.05 Å resolution is presented to facilitate the structure-based design of antiviral agents against ZIKV.
PDB reference: full-length Zika virus NS5 protein, 5tfr
FcpA from Leptospira interrogans, which is essential for pathogenesis, was crystallized but proved to be recalcitrant to X-ray diffraction studies. The orthologue from the saprophyte L. biflexa allowed three different crystal forms to be obtained that were suitable for crystallographic studies.
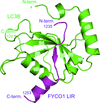
FYCO1 mediates microtubule plus-end-directed autophagosome transport by interacting with LC3. Here, the crystal structure of mouse LC3B in complex with the FYCO1 LC3-interacting region (LIR) motif peptide was determined.
PDB reference: LC3B in complex with FYCO1 LIR, 5wrd
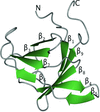
Structural analysis of two variant forms of truncated hemolysin A reveals additional proteolysis of the C-terminal 31 amino acids under high-salt crystallization conditions. This structural plasticity may support the implementation of stably exposed, on-edge β-strands during template-assisted folding.
The structure of ligand-free and DNA-unbound AmtR, a TetR-type global nitrogen regulator from C. glutamicum, was solved at 2.1 Å resolution in space group P21212 with six molecules in the asymmetric unit. A structure comparison with the previously solved structure of AmtR in a trigonal crystal system suggests a 6:6 stoichiometric association of three AmtR dimers with two trimeric GlnK complexes that prevents AmtR from binding to DNA and induces gene transcription.
PDB reference: AmtR, 5mqq
Cystathionine γ-synthase, a pyridoxal 5′-phosphate (PLP)-dependent enzyme from the thermoacidophilic archaeon S. tokodaii, was crystallized under three conditions. The structural data suggested that the orientations of the PLP cofactor and the active-site residues differed among the three forms, which might be key to the activity of the enzyme under extreme conditions.
A new molecular packing of pseudoazurin from A. faecalis shows a right-handed double helix composed of blue copper sites.
PDB reference: pseudoazurin, 5x31
Open  access
access
 access
accessThe structure of the C-terminal domain of CagX is presented and structural comparisons with TraO, its homologue from another bacterial T4S system, reveal distinct and conserved features.
PDB reference: CagX, 5h3v


 journal menu
journal menu














![[publBio]](/logos/publbio.gif)






