issue contents
August 2018 issue
Crystallography of glycoproteins and protein-carbohydrate complexes
Edited by Jon Agirre and Mark van Raaij
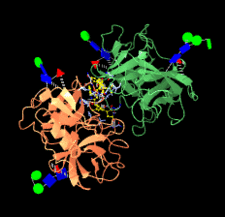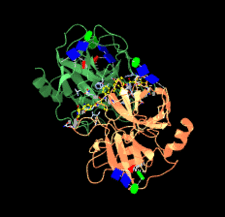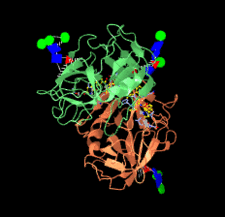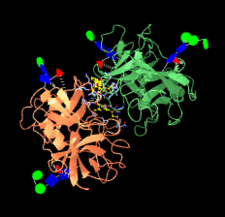
Cover illustration: Structure of highly glycosylated human leukocyte elastase in complex with an S2'-site binding inhibitor [Hochscherf et al. (2018), Acta Cryst. F74, 480-489]. Animation generated using the Glycoblocks representation [McNicholas & Agirre (2017), Acta Cryst. D73, 187-194] in CCP4mg.
editorial
An introduction to the Acta Cryst. F special issue on glycoproteins and protein–carbohydrate complexes in which the contents, the current state of the field and the future of glycan structural biology are briefly discussed.
topical reviews
Open  access
access
 access
accessProtein O-fucosyltransferases 1 and 2 perform the same post-transcriptional modification albeit on different substrates. Correct folding of the substrates is essential for protein recognition, and the crystal structures of these proteins have shown the mechanism of how these two proteins are highly specific.
Open  access
access
 access
accessProton-based saturation-transfer difference NMR (STD-NMR) is a technique that has proven to be particularly useful in the elucidation of protein–glycan interactions. This article explains the method for non-NMR spectroscopists and uses a number of crystal structures of viral capsid proteins in complex with respective glycan receptors to compare findings from crystallography and STD-NMR side by side, highlighting the complementary nature of the approaches.
research communications
Open  access
access
 access
accessThe results and challenges of carbohydrate handling in the current PDB-REDO databank are discussed.
Lacto-N-biose I units in the PDB were investigated. Instances and conformational features of the disaccharide in proteins and enzymes in the PDB from various organisms are reviewed.
A novel binding mode of a small-molecule inhibitor of human leukocyte elastase is revealed by its co-crystal structure with the enzyme. In the structure, a comparatively large part of the N-glycan chains attached to the enzyme are visible.
PDB reference: human leukocyte elastase, 6f5m
Open  access
access
 access
accessThe three-dimensional structure of a fungal arabinofuranosidase from CAZY family GH62 has been solved at 1.25 Å resolution in complex with the bespoke arabinofuranosidase inhibitor AraDNJ, shedding light on the activity of this catalyst in the enzymatic deconstruction of arabinoxylans.
Open  access
access
 access
accessThe CAZY family GH124 endoglucanase from Ruminiclostridium thermocellum contains an unusual metal-ion site featuring a covalently modified `2-oxohistidine' residue. Atomic resolution (∼1 Å) three-dimensional analyses, in conjunction with ITC and mass spectrometry, show that this metal site can accommodate a diverse array of transition metals, all of which confer a stabilizing effect on the enzyme.
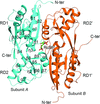
The structure of the effector-binding domain of CmpR in complex with the co-activator ribulose 1,5-bisphosphate is reported, revealing a novel ligand-binding pattern for the LysR-type transcriptional regulator family.
Open  access
access
 access
accessThe structure of a Vibrio cholerae `exo'-glycosidase from CAZy family GH9 has been solved at 3.17 Å resolution. Preliminary activity assays show that the enzyme is active towards both chitosan-derived oligosaccharides and β-glucosides.


 journal menu
journal menu














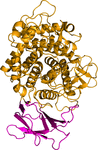
![[publBio]](/logos/publbio.gif)





