issue contents
September 2018 issue
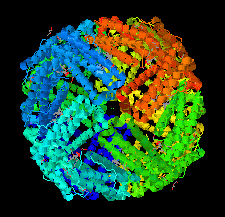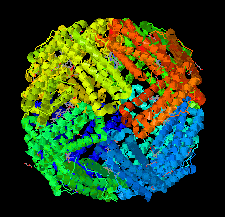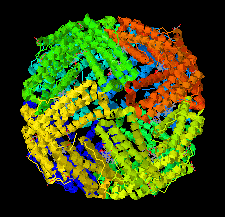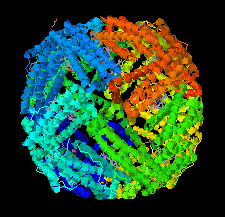
Cover illustration: The structure of bacterioferritin from Achromobacter at 1.95 Å [Dwivedy et al. (2018), Acta Cryst. F74, 558-566].
research communications
Open  access
access
 access
accessThe structure of the Fc fragment of the NIST reference antibody RM8671 is described in an orthorhombic crystal form. The molecular conformation is compared with those of precedents using a CH3-based reference frame and the pronounced asymmetry is linked to packing interactions.
PDB reference: Fc fragment of human IgG1 antibody from NISTmAb, 5vgp
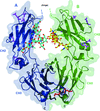
The crystal structure of a proteolytic fragment of an MeaB- and MMAA-like GTPase from Mycobacterium smegmatis was determined using the ab initio phasing program ARCIMBOLDO.
Open  access
access
 access
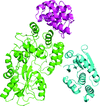
accessAnalysis of the noncrystallographic symmetry of crystals of the C-terminal domain of Burkholderia cenocepacia TssA indicates a quaternary structure of 32 subunits in D16 symmetry.
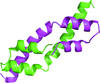
The Mycobacterium smegmatis Gre-factor homologue MSMEG_6292 is an RNA polymerase secondary channel-binding protein. To understand its structure and function, it was cloned, expressed, purified and crystallized, and crystallographic diffraction data were collected for both the native protein and a platinum derivative.
Open  access
access
 access
accessDesigned ankyrin-repeat proteins (DARPins) that bind to maltose-binding protein (MBP) with high affinity can facilitate the crystallization of an MBP fusion protein. The use of MBP-specific DARPins increases the probability of obtaining crystals.
This study reports the structure of bacterioferritin from Achromobacter sp. Dh1f, which was serendipitously crystallized.
PDB reference: Achromobacter sp. Dh1f bacterioferritin, 5zur
Staphylococcus aureus lipase (SAL), a possible target for anti-staphylococcal drugs, was expressed in Escherichia coli, purified and crystallized. A complete data set to 3.0 Å resolution was corrected and analyzed, suggesting a tetragonal space group (P4122 or P4322, a = b = 131.0, c = 250.6 Å) and four molecules per asymmetric unit.
The crystallization and X-ray diffraction analysis of the Cqm1 protein, which is known for its role as a receptor for mosquito-larvicidal binary toxin, is reported. The article also highlights the use of differential scanning fluorimetry for screening small-molecule ligands that could contribute to protein crystallization.
Open  access
access
 access
accessCrystals of different domain constructs of the TssA subunit from Aeromonas hydrophila have been crystallized in forms suitable for X-ray analysis.
The crystal structure of a non-immunosuppressive cyclic peptide inhibitor bound to its target, human cyclophilin A, is reported.
PDB reference: CypA–alisporivir complex, 5hsv
The binding of 28 dyes and stains to a dozen crystalline proteins, and to four in particular, was investigated by X-ray crystallography. Crystals of 31 different complexes were studied. Although firmly incorporated into the protein crystals in substantial amounts, the dyes are disordered and are possibly bound in a similar manner to conventional detergents.


 journal menu
journal menu

















![[publBio]](/logos/publbio.gif)





