issue contents
September 2019 issue
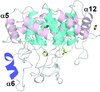
Cover illustration: A potential anti-infective drug target, GTP cyclohydrolase I from Listeria monocytogenes [Schüssler et al. (2019), Acta Cryst. F75, 586–592].
research communications
The structural analysis of a putative prenylcyclase enzyme, TchmY, from Actinoplanes teichomyceticus is reported.
PDB reference: TchmY, 6ij1
Open  access
access
 access
accessChe proteins are part of the signal transduction pathway between stimulus and response, mediated by the archaellum. The structure of the chemotaxis protein CheY was determined in two different crystal forms. The novel CheF protein that connects chemotaxis signaling to the motility apparatus was expressed and crystallized, and data were acquired by X-ray diffraction.
Open  access
access
 access
accessGTP cyclohydrolase I from Listeria monocytogenes, a putative anti-infective drug target, has been crystallized and the crystal structure was solved at 2.4 Å resolution.
PDB reference: GTP cyclohydrolase I, 4uqf
Stimulator of interferon genes (STING) is a key component of innate intracellular immunity. It is activated by the binding of cyclic dinucleotides (CDNs) produced by cyclic GMP-AMP synthase or of bacterial origin. The interactions of CDNs with STING are well understood; however, contrary to the previously published structure, it is shown that no Mg atoms are present in the binding site of STING.
PDB reference: STING in complex with 3′,3′-c-di-GMP, 6s86
The crystal structure of aminoglycoside 7′′-phosphotransferase-Ia from Streptomyces hygroscopicus in complex with hygromycin B has been determined at 2.4 Å resolution.
PDB reference: aminoglycoside 7′′-phosphotransferase-Ia, 6iy9
The crystal structure of UDP-glucose pyrophosphorylase from Yersinia pestis has been determined to 2.17 Å resolution, providing a foundation for future structure-based drug-design studies.
PDB reference: Y. pestis UDP-glucose phosphorylase, 6mnu
Open  access
access
 access
accessDihydrolipoamide succinyltransferase, a protein crystallization contaminant, was crystallized in a new crystal form in space group I4 and its structure was determined.
PDB reference: dihydrolipoamide succinyltransferase, 6pbr

 journal menu
journal menu












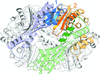

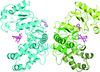
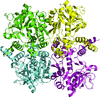

![[publBio]](/logos/publbio.gif)




