issue contents
December 2021 issue
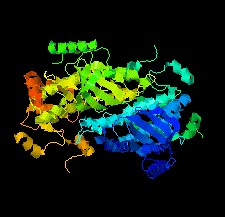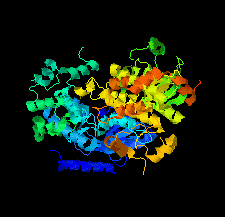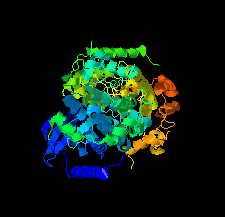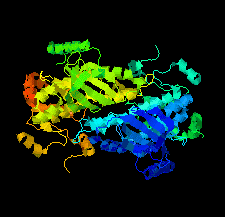
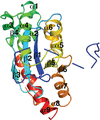
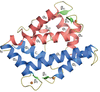
Cover illustration: Structure of mitogen-activated protein kinase kinase 1 in the DFG-out conformation [Nakae et al. (2021), Acta Cryst. F77, 459–464]. Eukaryotic protein kinases contain an Asp-Phe-Gly (DFG) motif, the conformation of which is involved in controlling the catalytic activity and can be switched between active-state (DFG-in) and inactive-state (DFG-out) conformations. The mechanism of conformational change is poorly understood, partly because there are few reports of the DFG-out conformation. Here, a novel crystal structure of nonphosphorylated human mitogen-activated protein kinase kinase 1 complexed with ATP-γS is reported in which MEK1 adopts the DFG-out conformation.
editorial
 access
accessresearch communications
 access
access

 journal menu
journal menu













![[publBio]](/logos/publbio.gif)





