issue contents
January 2022 issue
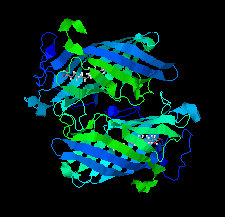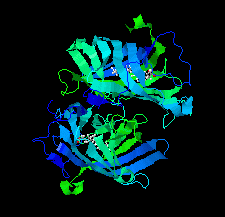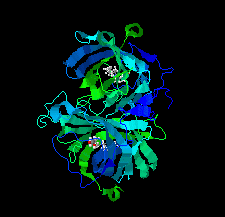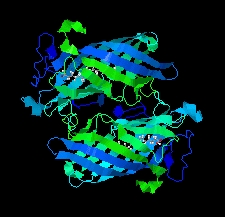
Cover illustration: DynF from the dynemicin-biosynthesis pathway of Micromonospora chersina. Access to the biosynthetic gene cluster of dynemicin, an enediyne natural product, has enabled the in vitro study of gene products within the cluster to decipher their roles in assembling this unique molecule. The crystal structure of one of these gene products, DynF, is reported by Kosgei et al. [(2022), Acta Cryst. F78, 1–7].
research communications
 access
access access
access access
access access
access

 journal menu
journal menu













![[publBio]](/logos/publbio.gif)





