issue contents
August 1997 issue
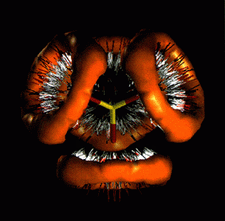
Cover illustration: Contoured distribution of hydrogen bonds from N-H and O-H donors about the oxygen acceptors of the disulfate dianion. Data for the 232 hydrogen bonds (O...H > 2.62 Å) were computed from the Cambridge Structural Database and the required symmetry was applied to generate the final distribution. Courtesy of M. Verdonk and J. Cole
topical reviews
This review takes a look at some of the strategies that have been devised for research in crystal engineering, in particular, those based around hydrogen-bonded architectures. In addition, some of the history and possibilites of the field itself are alluded to.
research papers
Download citation


Download citation


A structural model of the orthorhombic approximant (Imm2, a = 12.34, b = 12.41, c = 30.71Å) of the Al12Fe2Cr decagonal quasicrystal has been derived from its high-resolution electron microscopic images.
Download citation


Download citation


K3Na(MoO4)2 and K2.5Na1.5(MoO4)2 are monoclinic distorted glaserite structures. The excessive Na in K2.5Na1.5(MoO4)2 occupies the position of a more firmly bound potassium in the structure.
Download citation


Download citation


Analysis of total neutron scattering data has enabled us to determine the correct local structures in Li2MoO3 and Li4Mo3O8 – two compounds containing Mo–Mo-bonded triangular clusters. The shortcomings of analysing only Bragg scattering in the structure determination of disordered materials are demonstrated.
Download citation


Download citation


Crystals of pentachlorotoluene are, like crystals of most other hexasubstituted chloromethylbenzenes, rotationally disordered. Compositional disorder may also be a problem.
In Bridgman-grown monocrystals of CuInSe2, it has been found that the main cleavage planes are (101) and (112) and that twinning occurs along {112} planes about a [221] axis.
Download citation


Download citation


The crystal structure of the ionic conductor Li+/Na+ β-alumina has been studied at 30 and 298K with single-crystal X-ray diffraction and at 295K with neutron diffraction. The results reveal a complex ionic distribution of Li+ and Na+ ions.
Download citation


Download citation


High-temperature phases of Zn(ClO4)2.6H2O and Ni(ClO4)2.- 6H2O, two pseudohexagonal crystals with three orthorhombic twin components, have been studied by X-ray diffraction. Correlated disorder after temperature cycling has disappeared.
Download citation


Download citation


The crystal structure of the AgClO4.C6H6 complex, determined earlier by X-ray diffraction at room temperature, has been redetermined at 18, 78 and 158 K by neutron diffraction. Distortions of the benzene molecule from D6h symmetry ascribed to Ag+⋯C6H6 interactions are small, yet still significant.
Download citation


Download citation


Dimeric copper(II) benzoylformates show unusually strong antiferromagnetic interactions (-2J = ca 650 cm−1; H= -2JS1.S2), which may be due to the electronic effect of the α-keto group in the bridging carboxylate moiety. In the binuclear cage structure the coordination geometry around CuII atoms is typical square pyramidal.
Download citation


Download citation


Variable-temperature X-ray structure analysis of azobenzenes reveals that the torsional vibration of the N—Ph bonds is large enough to give rise to conformational interconversion in crystals.
The diffuse scattering analysis of the α-cyclodextrin cyclopentanone hydrate has revealed a variety of disorder phenomena; the interpretation of the results (structure in domains, intercell, vacancies and twin lattices) helps to explain the rotational disorder of the guest and some observed strains of the average structure. The correlated disorder phenomena are limited to small domains (with dimensions 102 or 103Å); the description of these domains suggests how crystals grow.
The frequency of formation and geometries of R1R2C=S⋯H-N,O systems show that these hydrogen bonds are resonance induced; the effective electronegativity of S is increased by conjugative interactions between C=S and substituent atoms (particularly N) or unsaturated groups at R1R2. Distance comparisons and acceptor `competition' studies involving >C=O⋯ H-N,O analogues show that C=S forms weaker hydrogen bonds than C=O.
The frequency of formation and the geometries of >S⋯H-N,O hydrogen bonds show that these bonds are both rare and very weak. Hydrogen bonds involving -S-H donors are, however, quite common, although weaker than analogous bonds involving O-H donors.
Download citation


Download citation


The crystal structures of pure L-carvone (C10H14O) and a DL-carvone equimolar mixture have been determined by Patterson-search methods at low resolution from powder diffraction data at 218 K. The mixed crystal formation in the chiral carvone system is confirmed, as proposed in recent thermodynamic studies.
Download citation


Download citation


The crystal packing of the diastereomeric mandelate salts of cinchonidine does not show the expected quasidiastereomeric relationship with the equivalent cinchoninium mandelate salts. The ineffectiveness of cinchonidine as a resolving agent for mandelic acid may be attributed to the isomorphism of the diastereomeric salts.
Download citation


Download citation


Crystallographic and ab initio studies have clarified the reason for yield variations in the reaction of differently substituted glutarimides with phenyllithium. It has been shown that conformational changes in glutarimide, introduced by substitution, resulted in electron charge distribution changes.
A survey of the positions of molecular centres in common space groups shows distinct patterns of preferred locations. There is a tendency for molecules to pack in patterns similar to ideal packing of spheres.
book reviews
Free 

Free 

books received
Free 

Free 



 journal menu
journal menu