issue contents
February 2014 issue
Special issue on Crystal engineering
Guest Editor: Professor Andrew D. Bond (University of Copenhagen, Denmark)
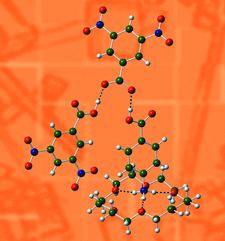
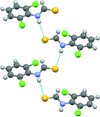
Cover illustration: Hydrogen-bonded motif in the crystal structure of a targeted ternary 4-aminobenzoic acid/crown ether/3,5-dinitrobenzoic acid complex. The crown ether is introduced specifically to sequester the NH3+ group of 4-aminobenzoic acid, leaving only the carboxylic acid group to interact with 3,5-dinitrobenzoic acid. See Fig. 5 in Boardman et al. [Acta Cryst. (2014), B70, 132-140].
editorial

feature articles

crystal engineering












Solvates of the antifungal drug griseofulvin: structural, thermochemical and conformational analysis
























international union of crystallography



 journal menu
journal menu