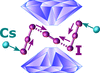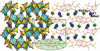issue contents
December 2021 issue
Cover illustration: Polyhedral representation of the crystal structure of trans-bis(oxalato)diaquarhodic acid tetrahydrate [see Garkul et al. (2021). Acta Cryst. B77, 1048–1054].
quantum crystallography
Free 

An overview is given by the guest editors of the papers published in the virtual special issue on quantum crystallography.
Open  access
access
 access
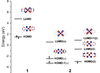
accessThe different nature of the emissive states of AgI and CuI one- and three-dimensional coordination polymers is here explained by a higher covalent character of the Cu—N bond with respect to the Ag—N one.
Download citation


Download citation


The transfer of theoretical multipole parameters to the experimental model was found to be helpful in separating the static electron density from the contaminant smearing effects emerging due to insufficiently accounted for atomic motion. A special refinement procedure resulted in significant improvement of the static electron density.
CCDC reference: 2084659
Download citation


Download citation


Open  access
access
 access
accessIntermolecular interactions of the new drug candidate sila-ibuprofen are investigated in the crystal and in the enzyme cyclooxygenase-II.
Open  access
access
 access
accessQuantum refinement has been shown to be a powerful approach to interpret and improve macromolecular crystal structures. Previous studies have shown that the results of quantum refinement can be improved if the charge of the quantum mechanical (QM) system is reduced by adding neutralizing groups. Here it is shown that a similar improvement can be obtained if the original highly charged QM system is instead immersed in a continuum solvent in the QM calculations.
Download citation


Download citation


Differences between the formal oxidation states of two nickel(II/III) coordination compounds with a non-innocent ligand were studied. There are differences between the theoretical calculations as well as between various experimental results.
scientific commentaries
Free 

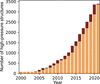
The remarkable growth in volume of high-pressure crystal structural chemistry depositions in the Cambridge Structure Database has been reported in a recent paper by Kaźmierczak & Patyk-Kaźmierczak [Acta Cryst. (2021), B77, 1012–1020].
Free 

A promising route towards rational design of materials is the combination of quantum chemical calculations with experimental electron density-based techniques within the framework of quantum crystallography. A recent work by Bartashevich et al. [Acta Cryst. (2021), B77, 478–487] fits well under the umbrella of modern QCr methods and it focuses on Appel's salt where, apart from purely covalent bonds, a variety of non-trivial intermolecular interactions are present.
high-pressure crystallography
Download citation


Download citation


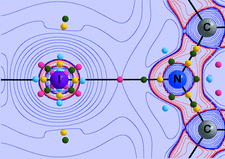
Iodine triiodide contacts in zigzag octaiodide units in Cs2I8 undergo substantial compression, down to intramolecular distances typical for covalently bonded polyiodides. The polyiodide net twists out of its initial shape, allowing for even more expanded connectivity between caesium cations and negatively charged iodine layers.
research papers
Download citation


Download citation


Meyerhofferite is known to undergo a pressure-mediated phase transition between 3.0 and 3.5 GPa. In this study, the crystal structure of the high-pressure polymorph of meyerhofferite and its pressure-induced deformation mechanisms are revealed, providing information aimed at improving knowledge of the high-pressure phase transitions in this class of hydrated borates.
Download citation


Download citation


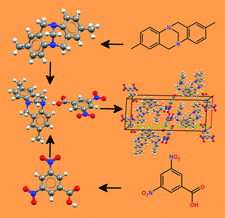
Eleven cocrystals along with a fourth polymorph of deferiprone are reported. The homo- and hetero-synthons preference amongst the cocrystal systems is discussed.
Download citation


Download citation


A detailed examination of intermolecular interactions via topological analysis of electron density and optical properties has been made of two polar materials containing the hydrochlorothiazide molecule.
Download citation


Download citation


Open  access
access
 access
accessStructural studies of 27 N-(methoxysalicylidene)-haloaniline (halo = F, Cl or Br) compounds are reported. New polymorphs were identified for two of the compounds and a phase transition was also identified for another. The link between their structures and visual observations of their thermochromic behaviour are made.
CCDC references: 2087834; 2087835; 2087836; 2087837; 2087838; 2087839; 2087840; 2087841; 2087842; 2087843; 2087844; 2087845; 2087846; 2087847; 2087848; 2087849; 2087850; 2087851; 2087852; 2087853; 2087854; 2087855; 2087856; 2087857; 2087858; 2087859; 2087860; 2087861; 2087863; 2087864; 2111142; 2111143
From an analysis of published results, it is concluded that the room-temperature structure of Ba2NaNb5O15 is that proposed in 1989 in the space group Bbm2 (Ama2 in standard setting). The structures and phase transitions at higher temperatures are also considered.
Download citation


Download citation


Open  access
access
 access
accessThe crystal structure of a previously unknown small heterocyclic alkanolamine was determined by X-ray powder diffraction. Total scattering analysis of small organic molecules is shown to be useful to help disambiguate spectroscopic and elemental analyses.
CCDC reference: 2061886
Download citation


Download citation


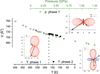
The phase transition observed in a temperature-dependent experiment at 174 K is unachievable under high-pressure conditions. Negative thermal expansion for phase (II) and negative compressibility for phase (I) were observed.
Download citation


Download citation


Synthesis, crystal structure and evaluation of a possible ion migration pathways are reported for a novel highly porous Na-K-Cu anhydrous sulfate. The overall coordination polyhedra of Cu2+ in the new compound can be considered as `octahedra with one split vertex'.
CCDC reference: 2114165
An in-depth analysis of the high-pressure entries of the Cambridge Structural Database is presented.
Download citation


Download citation


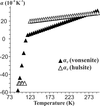
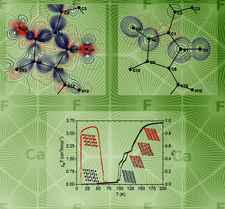
Vonsenite and hulsite were investigated by a set of in situ low-temperature methods. Cascades of magnetic transitions were revealed in both oxoborates and the critical temperatures were determined. Anomalies in the unit-cell parameters and thermal expansion of the oxoborates at critical temperatures were observed.
Download citation


Download citation


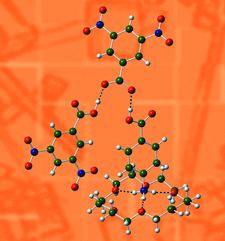
Experimental and theoretical charge density analysis of a cocrystal of nicotinic acid and pyrogallol provides insight into the mechanism of nucleation and growth of the cocrystal.
Download citation


Download citation


The polymorphism of Na[Rh(H2O)2(C2O4)2]·2H2O is noteworthy. The complex crystallizes with the formation of two different structures of the same water content. Oxalato-rhodic acid not only exists in solution, but, unlike most complex acids, it can also crystallize as a tetrahydrate.

 journal menu
journal menu