issue contents
September 2005 issue
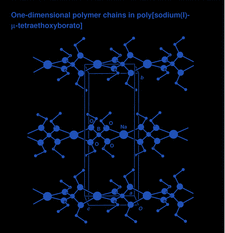
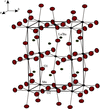
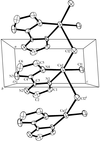
Cover illustration: The unit-cell contents of catena-poly[sodium(I)-![[mu]](/logos/entities/mu_rmgif.gif) -tetraethoxyborato], viewed along the a axis, showing the one-dimensional polymer chains along the twofold c axis. See Gainsford & Kemmitt [Acta Cryst. (2005), C61, m417-m418].
-tetraethoxyborato], viewed along the a axis, showing the one-dimensional polymer chains along the twofold c axis. See Gainsford & Kemmitt [Acta Cryst. (2005), C61, m417-m418].
inorganic compounds
Download citation


Download citation


La0.815Ba0.185MnO3 crystallizes with a monoclinic (I2/c) perovskite-type structure at 160 K.
Download citation


Download citation


InAsO4·H2O crystallizes in the framework structure type of MnMoO4·H2O and is characterized by In2O8(H2O)2 dimers of edge-sharing InO5(H2O) octahedra corner-linked to AsO4 tetrahedra. PbIn(AsO4)(AsO3OH) also has an octahedral–tetrahedral framework structure and is isotypic with PbFeIII(AsO4)(AsO3OH).
Download citation


Download citation


CsGa(H1.5AsO4)2(H2AsO4) and isotypic CsCr(H1.5AsO4)2(H2AsO4) represent a novel structure type containing decorated kröhnkite-like octahedral–tetrahedral chains and a very short hydrogen bond (O⋯O distance ∼2.51 Å) involving a split H-atom position.
Download citation


Download citation


AgHg3SbO6 adopts the rhombohedral K4CdCl6 structure type featuring chains of alternating face-sharing SbO6 and AgO6 octahedra along [001]. The octahedral chains are separated by Hg2+ cations in linear coordination.
Download citation


Download citation


La2RuO5 shows a phase transition from a monoclinic high-temperature to a triclinic low-temperature modification at approximately 160 K. Powder neutron diffraction at 293 and 1.5 K was used to study both structures.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu