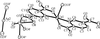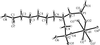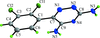issue contents
September 2009 issue
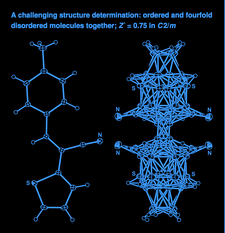
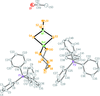
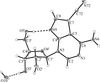
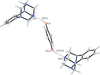
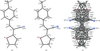
Cover illustration: The independent molecular components of (E)-3-(4-methylphenyl)-2-(2-thienyl)acrylonitrile, showing molecule 1 (left) and the four orientations of molecule 2 across a site of 2/m symmetry at (0, 0, ½) (right). Displacement ellipsoids are drawn at the 30% probability level. See Cobo, Quiroga, Cobo & Glidewell [Acta Cryst. (2009), C65, o444-o446].
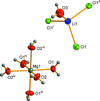
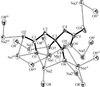
inorganic compounds




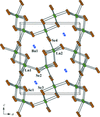
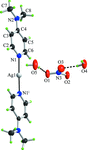
metal-organic compounds




















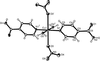
organic compounds


































 journal menu
journal menu