issue contents
October 2011 issue
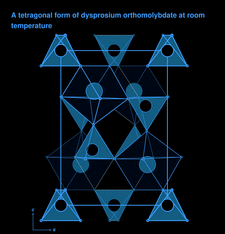
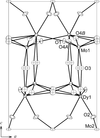
Cover illustration: The structural model of Dy2(MoO4)3, viewed along the b axis. Tetrahedra are formed by Mo atoms and polyhedra by Dy atoms. See Dorzhieva, Chumak, Sarapulova, Mikhailova, Bazarova & Ehrenberg [Acta Cryst. (2011), C67, i50-i52].
inorganic compounds
Download citation


Download citation


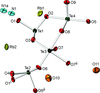
The crystal structure of ammonium rubidium nonaoxotetratellurate(IV) dihydrate has been studied as a function of pressure up to 7.40 GPa. No phase transition is observed.
Download citation


Download citation


The single crystals of the tetragonal modification of dysprosium orthomolybdate obtained in the present work are stable at room temperature. The crystal structure is formed by corner-sharing MoO4 tetrahedra and Dy polyhedra coordinated by seven O atoms.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu