issue contents
January 2011 issue

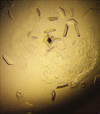
Cover illustration: A selection of the best crystal pictures published in Acta Cryst. F in 2010.
editorial
Free 

structural communications
Three crystal structures of the molybdenum-cofactor biosynthesis protein MogA from two highly thermophilic organisms have been determined at high resolution. Comparative analyses revealed the residues involved in oligomerization. In addition, molecular-dynamics and docking studies suggested the binding affinities of several small molecules towards MogA and homologous proteins.
The crystal structure of SEp22, a DNA-binding protein from starved cells from S. enterica subsp. enterica serovar Enteritidis, was determined in two forms: the native state at 1.25 Å resolution and an iron-soaked form at 1.30 Å resolution.
Open  access
access
 access
accessThe structure of the C-terminal domain of the S. mutans surface adhesin SpaP has been determined to 2.2 Å resolution.
PDB reference: C-terminal domain of SpaP, 3opu
The structure of the small laccase from S. coelicolor is reported at improved resolution and in a different space group. The soaked ligand is bound between laccase molecules.
PDB reference: laccase, 3kw8
Open  access
access
 access
accessThe structure of L. donovani pteridine reductase has been targeted to assist in a program of structure-based inhibitor research. Crystals that diffracted to 2.5 Å resolution were obtained and the structure has been solved. Unfortunately, the active site is disordered and this crystal form is unsuitable for use in characterizing enzyme–ligand interactions.
PDB reference: pteridine reductase, 2xox
crystallization communications
H. sapiens quinolinate phosphoribosyltransferase has been expressed, purified and crystallized. A diffraction data set has been collected and processed at 2.8 Å resolution.
Crystals of native intimin and its N916Y mutant from enterohaemorrhagic E. coli O157:H7 diffracted to 2.8 and 2.6 Å resolution, respectively.
Bacterial blight, an infectious disease caused by X. oryzae pv. oryzae (Xoo), causes huge rice-production losses in most rice-cultivating countries. The co-chaperonin of Xoo, XoGroES, an important protein for protein folding, was cloned from Xoo, purified and crystallized for atomic resolution structure determination.
The human G3BP1 NTF2-like domain was crystallized. Diffraction data were collected to 3.6 Å resolution.
Crystals of the thiaminase type II from S. aureus are orthorhombic, belonging to space group P212121 with unit-cell parameters a = 103.5, b = 104.1, c = 109.6 Å, and diffracted to 2.6 Å resolution.
Human α-thrombin was crystallized in complex with specific peptide inhibitors of general sequence D-Phe-Pro-D-Arg-P1′-CONH2. The crystals belonged to the orthorhombic space group P212121 and diffracted to beyond 1.3 Å resolution.
Biphenyl 2,3-dioxygenase from B. xenovorans LB400 and its variants BPDOP4 and BPDORR41 were crystallized using agarose gel and the crystals were characterized using X-ray diffraction.
The vWA domain of human anthrax toxin receptor 1 was overexpressed in E. coli, purified and crystallized. Diffraction data were collected to 1.8 Å resolution.
α-Glucuronidase from S. pristinaespiralis was crystallized by the sitting-drop vapour-diffusion method. The crystals belonged to space group R3 and diffracted to a resolution of 1.9 Å.
The cloning, expression, purification, crystallization and preliminary X-ray analysis of wild-type and of an active-site mutant of C. jejuni glyceraldehyde-3-phosphate dehydrogenase is reported.
SMU.1108c, a putative uncharacterized protein from S. mutans, was crystallized and X-ray diffraction data were collected to a resolution of 2.2 Å.
The purification, crystallization and preliminary X-ray diffraction analysis of RNase HIII from S. aureus is presented. Crystals that diffracted to 2.6 Å resolution in space group P212121 were only obtained after removal of the hexahistidine tag.
ESX-1 secreted protein regulator (EspR, Rv3849) from M. tuberculosis has been purified and crystallized, and diffracted to 3.2 Å resolution at wavelength 0.97625 Å.
Crystallization of basic 7S globulin from soybean is presented.
An apo form of human arginase I which is suitable for soaking experiments has been crystallized.
In order to investigate its structure and function, the NmrA-like domain-containing DDB_G0286605 protein from D. discoideum was expressed, purified and crystallized. X-ray diffraction analysis is reported to a resolution of 1.64 Å.
In order to investigate its structure and function, the NmrA-like short-chain dehydrogenase/reductase-type DDB_G0291732 protein from D. discoideum was expressed, purified and crystallized. X-ray diffraction analysis is reported to a resolution of 1.65 Å.
Isopentenyl diphosphate isomerase from M. jannaschii has been overexpressed in E. coli, purified and crystallized. Diffraction data were collected to 2.08 Å resolution.
The small terminase subunit of bacteriophage P22 was overexpressed in E. coli, purified under native conditions and crystallized as a nonamer. High-quality diffraction data were collected to 1.75 Å resolution using synchrotron radiation.
A β-glucosidase A (BglA) from the thermophile Halothermothrix orenii has been cloned, purified and crystallized in an orthorhombic space group. X-ray diffraction data have been collected to 3.5 Å resolution, and the structure was solved by molecular replacement, revealing the presence of two molecules in the asymmetric unit.
The crystallization of E. coli maltoporin in a new crystal form that diffracts to high resolution is reported.
Wild-type and a kinase-dead mutant of the C-terminal serine/threonine kinase domain of CTR1 from A. thaliana were expressed and crystallized. The crystals belonged to space groups P41212 and P42212, respectively.
PpAzoR, an FMN-dependent NADPH azoreductase from Pseudomonas putida MET94, has been crystallized using the sitting-drop vapour-diffusion technique.
Glycosylated recombinant bifunctional nuclease from tomato has been crystallized and preliminary X-ray diffraction analysis was performed.
LR11/sorLA contains in its extracellular region a large (∼700-residue) Vps10p domain that is implicated in its intracellular protein-trafficking function. Here, the expression, purification, crystallization and preliminary crystallographic characterization of this domain are described.
NifH2, a homologue of nitrogenase reductase from Methanocaldococcus jannaschii was cloned, overexpressed, purified and crystallized. X-ray diffraction data were collected to 2.85 Å resolution and the crystals belonged to space group P2.
The recombinant glycosyltransferase ElaGT from the elaiophylin-producing marine Streptomyces sp. SCSIO 01934 has been overexpressed in E. coli, purified and crystallized. Diffraction data were collected to 2.9 Å resolution.
A variant of the diaminopimelate/lysine pathway has recently been defined following the discovery of the enzyme L,L-diaminopimelate aminotransferase (DapL). The cloning of the cDNA, recombinant expression, purification and preliminary diffraction analysis of DapL from the alga C. reinhardtii are presented.
The KaiC-like protein PH0187 from the hyperthermophilic archaeon P. horikoshii OT3 was expressed, purified and crystallized using the sitting-drop vapour-diffusion method. The crystal of PH0187 diffracted X-rays to 2.75 Å resolution.
D-Serine dehydratase purified from chicken kidney was crystallized by the hanging-drop vapour-diffusion method. The crystals diffracted to 2.09 Å resolution.
The crystallization and preliminary X-ray analysis of a cold-active endo-β-1,4-D-xylanase is described. The crystals diffracted to 2.7 Å resolution.
The seryl-tRNA synthetase from C. albicans was crystallized by the sitting-drop vapour-diffusion method using ammonium sulfate as precipitant. The crystals belonged to the hexagonal space group P6122 and diffraction data were collected to 2.0 Å resolution at a synchrotron source.
Limited proteolysis of a monomeric fraction of Tellina virus 1 VP4 protease leads to crystals that diffract to beyond 2.1 Å resolution.
Crystals of the lytic transglycosylase MltE from E. coli were grown using the microbatch method and diffracted to a resolution of 2.1 Å.
In this study, the region 115–362 of the resuscitation-promoting factor RpfB was cloned, expressed and crystallized. This region includes the C-terminal catalytic domain, the G5 domain and one of the three DUF348 domains, which are of previously unknown structure and function.
The genome sequence of mimivirus, the largest known double-stranded DNA virus, encodes a putative protease: the R355 gene product. Its expression in E. coli, its crystallization and the preliminary phasing of a MAD data set using the selenium signal present in a crystal of recombinant selenomethionine-substituted protein are reported.
international union of crystallography
Free 



 journal menu
journal menu




























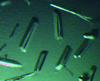
















![[publBio]](/logos/publbio.gif)





