issue contents
January 2014 issue

Cover illustration: A selection of crystals from articles published in 2013.
editorial
Free 

The Editors highlight the crystals featured on the cover and a new series of articles.
IYCr crystallization series
The gateway to X-ray crystallography is the growth of macromolecular crystals. An attempt is made here to acquaint investigators with the principles and practice of protein crystallization, some recent innovations, and to touch upon some of the remaining problems.
Structural and biochemical data demonstrate that p202 recognizes foreign DNA in a distinct manner compared with AIM2/Aim2, leading to a new model for the negative regulation of Aim2-mediated inflammasome activation by p202.
PDB reference: p202 HINa domain, 4lnq
An oligopeptide containing a reduced analogue of a Phe-Tyr dipeptide was found to have good inhibition activity against endothiapepsin, and the crystal structure of the inhibitor–enzyme complex has been determined at 1.35 Å resolution.
PDB reference: endothiapepsin–FRY complex, 4lp9
The structures of T. brucei brucei adenosine kinase in open and closed conformations give insight into the substrate-binding mode and can be used to improve the properties of antitrypanosomal agents.
The crystal structure of the R80N mutant of nucleoside diphosphate kinase from M. tuberculosis has been determined at 1.9 Å resolution. Biochemical data and structural analysis provide important information on the stability of the hexamer. A citrate is bound in the catalytic site where the nucleotide phosphate groups are usually located.
PDB reference: nucleoside diphosphate kinase, R80N mutant, 4ane
The DNA-binding domains of Ets1 and FOXO1 were expressed, purified, and crystallized in complex with DNA containing a composite sequence for a noncanonical forkhead binding site and an ETS site. Diffraction data were collected to a resolution of 2.2 Å.
The expression, purification, crystallization and preliminary X-ray diffraction analysis of a novel keto-deoxy-D-galactarate dehydratase from A. tumefaciens are described.
The cloning, overexpression, purification, crystallization and preliminary X-ray crystallographic analysis of phosphoglycerate mutase from S. aureus NCTC8325 are reported.
The expression, purification, crystallization and preliminary crystallographic analysis of the high-mobility group protein (HMO2) from S. cerevisiae are reported.
The crystallization and preliminary X-ray study of a complex of uridine phosphorylase from V. cholerae with 6-methyluracil are reported.
The crystallization and preliminary X-ray characterization of an HA17–HA20 complex from C. botulinum type C toxin are reported.
The N-terminal calmodulin-like domain of the human mitochondrial ATP-Mg/Pi carrier SCaMC1 was crystallized in the presence of Ca2+. X-ray diffraction data were collected to 2.9 Å resolution from crystals which belonged to space group P6222.
A double mutation (P453H and I471T) improved the solubility of the middle domain of PB2 from influenza A virus and the shape of the crystals that were obtained. A small crystal was used to test the diffraction and a preliminary X-ray diffraction data set was collected to 2.30 Å resolution.
Crystals of the small subunit of POXA3b, a heterodimeric laccase (multicopper oxidase) from P. ostreatus, belonged to space group P41212 or P43212 and contained two molecules in the asymmetric unit related by a noncrystallographic twofold axis. X-ray data were collected to a maximum resolution of 2.5 Å using a synchrotron source.
The structure of mouse protein arginine methyltransferase 7 was solved by the SAD method. The protein was crystallized in two different crystal forms.
The DNA-binding protein BurrH from B. rhizoxinica has been expressed, purified and crystallized in its apo form and in complex with its target DNA. Preliminary X-ray diffraction analysis is reported.
The preparation and crystallographic study of S. japonicum thioredoxin glutathione reductase are reported.
A feast/famine regulatory protein (Rv2779c) from M. tuberculosis H37Rv was cloned, overexpressed, purified and crystallized. The crystals diffracted to a resolution of 2.8 Å and belonged to space group P21212.
β-Ketoacyl-(acyl carrier protein) reductase (FabG), an enzyme from Y. pestis involved in fatty-acid synthesis, was crystallized and X-ray diffraction data were collected to 2.3 Å resolution.
Recombinant XynAS9, a thermophilic GH10 xylanase from Streptomyces sp. S9, was crystallized and X-ray diffraction data were collected to 2.08 Å resolution.
L-Arabinose isomerase from G. kaustophilus was overexpressed, purified and crystallized, and a preliminary X-ray diffraction data set was obtained.
OsRac1, a small GTPase from O. sativa, was expressed, purified and crystallized. An X-ray diffraction data set was collected from a native crystal to 1.9 Å resolution.
Mammalian mitochondrial Hsp60 and Hsp10 chaperonins were expressed, purified and co-crystallized. The structure of their symmetrical complex was determined by molecular replacement.
Recombinant human fumarase has been overexpressed in E. coli and crystallized in the presence of polyethylene glycol as a precipitant agent. The structure was solved by molecular-replacement techniques.
Despite possessing an N-terminal F-BAR domain, the srGAP2 protein regulates neurite outgrowth and neuronal migration by causing membrane protrusions reminiscent of the activity of I-BAR domain proteins. In this study, the F-BAR domain of human srGAP2 was crystallized and diffracted to 3.50 Å resolution.
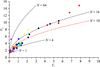
The intrinsic variability between and within macromolecular crystals has a large effect on screening and data-collection strategies. Measures of this variability and their implications for data collection are presented.


 journal menu
journal menu
































![[publBio]](/logos/publbio.gif)





