issue contents
August 2014 issue
Cover illustration: The first structure of the catalytic domain of RpfC (Rv1884), one of the resuscitation-promoting factors (RPFs) from Mycobacterium tuberculosis (Chauviac et al., p. 1022).
IYCr crystallization series
Open  access
access
 access
accessAs technology advances, the crystal volume that can be used to collect useful X-ray diffraction data decreases. The technologies available to detect and study growing crystals beyond the optical resolution limit and methods to successfully place the crystal into the X-ray beam are discussed.
structural communications
In this study, the crystal structure of Saccharomyces cerevisiae mitochondrial Qri7 complexed with AMP (a part of threonylcarbamoyl-adenylate) has been determined at 2.94 Å resolution in a new crystal form.
PDB reference: Qri7, 3wuh
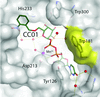
The structure of AA(Gly)B, a dual-specificity blood group synthase, in complex with UDP-galactose, the O6′′ atom of which is protected by an 2-nitrobenzyl group, has been determined in two space groups. The so-called caged substrate exhibits various distinct conformations in the active site.
Open  access
access
 access
accessThe structure of the catalytic domain of RpfC (Rv1884) closely resembles those of RpfB and RpfE. This structural conservation highlights the importance of the versatile domain composition of the RPF family.
PDB reference: RpfC, 4ow1
The crystal structure of the human RIG-I helicase domain, a key player protein in the antiviral innate immune response, is reported.
PDB reference: RIG-I helicase domain, 4on9
The α chain of haemoglobin (α) has been crystallized as a homodimer bound to a domain from the S. aureus haemoglobin receptor iron-regulated surface determinant H (IsdH). This is the first X-ray crystal structure that captures the weak self-association interface of α.
crystallization communications
A GH127 β-L-arabinofuranosidase from G. stearothermophilus T6 (Ara127N) has been purified and crystallized in two different crystal forms. A full X-ray diffraction data set was measured for the wild-type enzyme in a P212121 crystal form (2.3 Å resolution) and complete MAD data were measured for the selenomethionine derivative in a P21 crystal form (2.5 Å resolution) to be used for the detailed three-dimensional structural analysis of the Ara127N protein.
D-Lactate dehydrogenase from L. jensenii (LjD-LDH), a key enzyme for the production of the D-form of lactic acid from pyruvate, has been crystallized and X-ray diffraction data were collected from the crystal and analyzed.
The crystallization of a putative food allergen from pecan is reported.
The human TLR6 TIR domain was crystallized and crystals were found to belong to space group C2, with unit-cell parameters a = 127.60, b = 44.20, c = 75.72 Å, β = 118.89°. The crystals were obtained at 20°C and diffracted to a resolution of 2.2 Å.
Open  access
access
 access
accessYidC, a membrane-protein chaperone/insertase from B. halodurans, was expressed, purified and crystallized in the lipidic cubic phase. An X-ray diffraction data set was collected to 2.4 Å resolution.
The R. flavefaciens scaffoldin C cohesin complexed with the dockerin from a CBM-containing protein has been crystallized. Data were collected to 2.5, 2.4 and 2.16 Å resolution from three different crystalline forms.
The fourth scaffoldin A cohesin complexed with the dockerin of a family 5 glycoside hydrolase from A. cellulolyticus has been purified and crystallized, and data were processed to a resolution of 1.57 Å in space group P212121.
This article describes preliminary crystallographic data of a complex consisting of human coronavirus NL63 main protease and its inhibitor N3.
X-ray diffraction data for Amphi-IgSF-V (a single variable domain of the immunoglobulin superfamily from amphioxus) were collected to 1.95 Å resolution. The crystal belonged to space group P3221, with unit-cell parameters a = b = 53.9, c = 135.5 Å, and contained two molecules in the asymmetric unit. The Matthews coefficient and solvent content were calculated to be 2.58 Å3 Da−1 and 52.38%, respectively.
A single-strand-specific 3′–5′ exonuclease, PhoExo I, from the hyperthermophilic archaeon P. horikoshii OT3 was produced as inclusion bodies in E. coli cells, solubilized by the high-pressure refolding method, purified and crystallized. The crystal of PhoExo I diffracted X-rays to 1.52 Å resolution.
This is the first report of the crystallization conditions for VLPs from a grouper fish nervous necrosis virus, and the VLP structure diffracted X-rays to 7.5 Å with high completeness.
The N-D1 domain of human VCP was crystallized in complex with the N domain of yeast OTU1 and X-ray diffraction data were collected to 3.25 Å resolution.
Open  access
access
 access
accessThe cloning, expression, purification, crystallization and preliminary X-ray diffraction analysis of Rv3705c from M. tuberculosis are described.
The human Uba5 C-terminal domain was demonstrated to be necessary for formation of the Ufc1–Ufm1 complex, and the minimal region for Ufc1 recognition was identified. The fusion protein between Uba5 381–404 and Ufc1 was expressed, purified and crystallized, and diffraction data were collected to 3.0 Å resolution.
A ribokinase from V. cholerae O395 has been cloned, overexpressed, purified and crystallized both in a native form and in a ribose- and ADP-bound form. Both crystals belonged to space group P1. Apo crystals of ribokinase diffracted to 3.4 Å resolution and the co-crystals diffracted to 1.75 Å resolution.
The C-terminal coiled-coil (CC3) region of mouse Bicaudal-D1 (BICD1) and its complex with the small GTPase Rab6 were purified and crystallized. Crystals of the BICD1 CC3 region alone were obtained, but not of the BICD1–Rab6 complex, and X-ray diffraction data were collected from the BICD1 CC3 crystals to a resolution of 1.50 Å for phase determination by the multi-wavelength anomalous dispersion method.
laboratory communications
Open  access
access
 access
accessProtein crystallization with microseed matrix screening: application to human germline antibody Fabs
The power of microseed matrix screening is demonstrated in the crystallization of a panel of antibody Fab fragments.


 journal menu
journal menu












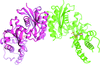
















![[publBio]](/logos/publbio.gif)





