issue contents
July 2015 issue
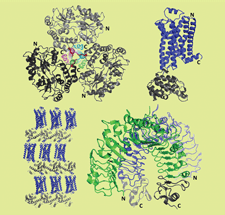
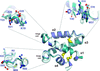
Cover illustration: Examples of the successful application of the heterologous fusion-protein approach to crystallization. See Kobe et al. (p. 861) for details.
ICCBM15
Free 

Selected papers from ICCBM15 have been published in the July 2015 issue of Acta Crystallographica Section F, Structural Biology Communications.
Open  access
access
 access
accessThe presence of a covalently bound fluorescent probe at a concentration of <0.5% does not affect the outcome of macromolecule crystallization screening experiments. Additionally, the fluorescence can be used to determine new, not immediately apparent, lead crystallization conditions.
Open  access
access
 access
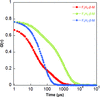
accessThe evolution of protein-rich clusters and nucleating crystals were characterized by dynamic light scattering (DLS), confocal depolarized dynamic light scattering (cDDLS) and depolarized oblique illumination dark-field microscopy. Newly nucleated crystals within protein-rich clusters were detected directly. These observations indicate that the protein-rich clusters are locations for crystal nucleation.
An X-ray compatible microfluidic crystallization platform enables in situ time-resolved serial Laue crystallography.
Open  access
access
 access
accessMORPHEUS II is a 96-condition initial crystallization screen formulated de novo. The screen incorporates reagents selected from the Protein Data Bank to yield crystals that are not observed in traditional conditions. In addition, the formulation facilitates the optimization and cryoprotection of crystals.
The influence of fluorinated segment length on surfactant micelle form factors, intermicellar interaction parameters and the surfactant phase diagram in solution conditions for membrane-protein crystallization is reported.
The novel thermostable aldo-keto reductase Tm1743 from T. maritima was overexpressed with an N-terminal His6 tag, purified and co-crystallized with NADP+. Degradation of the N-terminal vector-derived amino acids was identified by Western blot and mass-spectrometric analyses.
Open  access
access
 access
accessA new batch preparation method is presented for high-density micrometre-sized crystals of the G protein-coupled receptor rhodopsin for use in time-resolved serial femtosecond crystallography at an X-ray free-electron laser using a liquid jet.
This review describes the two main applications of fusion proteins in protein crystallization: the `heterologous fusion-protein approach' and the `fusion of interacting proteins approach'.
research communications
A. thaliana BAG5 (AtBAG5) and calmodulin were expressed and purified separately and then co-crystallized. The preliminary X-ray diffraction studies of the protein complex are reported. Structure determination will ultimately provide insights into the mode of interaction between AtBAG5 and calmodulin.
Open  access
access
 access
accessThe crystal structures of the individual domains of the Mex67–Mtr2 complex from C. thermophilum have been determined and their arrangement in solution has been studied by SAXS.
PDB references: Mex67 RRM, 4wpm; Mex67 UBA, 4wp2; Mex67 NTF2L, 4wp5; Mex67 LRR, 4wp6; Mtr2, 4x2m; Mex67 LRR+NTF2L, 4xm4
A novel aromatic prenyltransferase from A. terreus, named AtaPT, was found to prenylate diverse novel aromatic compounds. The expression and crystallization of AtaPT are reported here.
Open  access
access
 access
accessA 1.8 Å resolution structure of the sphingolipid activator protein saposin A has been determined at pH 4.8, the physiologically relevant lysosomal pH for hydrolase enzyme activation and lipid-transfer activity.
PDB reference: saposin A, 4uex
The C-terminal sterol carrier protein type 2 (SCP-2) domain of human hydroxysteroid dehydrogenase-like protein 2 (including residues Lys318–Arg416) was produced in E. coli, purified and crystallized, and X-ray diffraction data were collected to 2.10 Å resolution.
The crystal structure of mouse nerve growth factor (NGF) complexed with lysophosphatidylinositol was solved and the structural determinants of NGF for LysoPI molecule recognition were identified. The influence of LysoPI in the interactions between NGF and its two receptors is modelled.
PDB reference: nerve growth factor–lysophosphatidylinositol complex, 4xpj
The expression, purification and crystal structure of a recombinant fluorobody to TLH are reported.
PDB reference: fluorobody, 4xgy
YdiE, a conserved protein apparently involved in haem transport, is one of the smallest proteins produced by E. coli at only 63 residues in length. The N-terminal 20 residues of each chain in the homodimer were disordered, but crystals of the full-length protein allowed structure refinement to 1.5 Å resolution.
PDB reference: YdiE, 4ynx
Open  access
access
 access
accessExpression, purification, crystallization and crystallographic study of Lutzomyia longipalpis LJL143
LJL143, a salivary protein from L. longipalpis, was produced using P. pastoris and crystallized in space group P212121.


 journal menu
journal menu














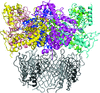


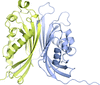





![[publBio]](/logos/publbio.gif)






