issue contents
January 2020 issue
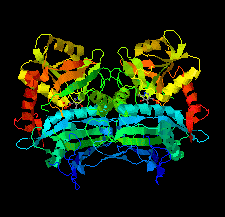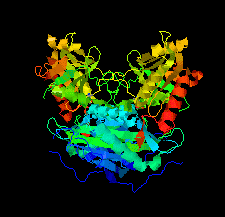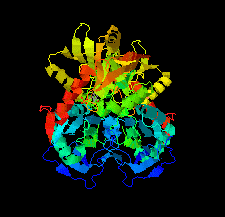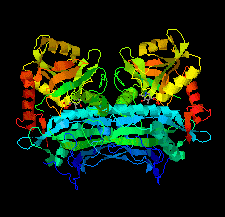
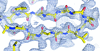
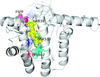
Cover illustration: An oxidized mutant of human mitochondrial branched-chain aminotransferase (hBCAT) [Herbert et al. (2020), Acta Cryst. F76, 14-19]. hBCAT catalyzes the transamination of the branched-chain amino acids leucine, valine and isoleucine and ![[alpha]](/logos/entities/alpha_rmgif.gif) -ketoglutarate to their respective
-ketoglutarate to their respective ![[alpha]](/logos/entities/alpha_rmgif.gif) -keto acids and glutamate. This enzyme thus plays a significant role in amino-acid metabolism and whole-body nitrogen shuttling, in particular with respect to the de novo synthesis of the neurotransmitter glutamate in the brain.
-keto acids and glutamate. This enzyme thus plays a significant role in amino-acid metabolism and whole-body nitrogen shuttling, in particular with respect to the de novo synthesis of the neurotransmitter glutamate in the brain.
research communications
 access
access

 journal menu
journal menu
















![[publBio]](/logos/publbio.gif)





