issue contents
March 2020 issue
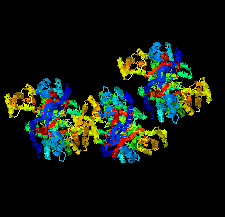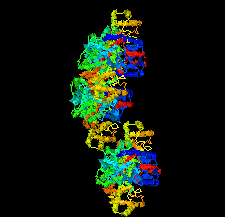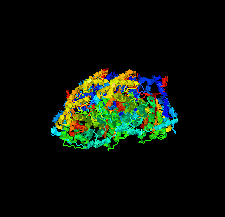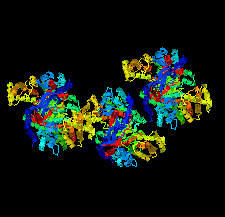
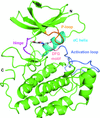
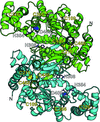
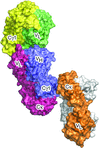
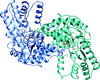
Cover illustration: Structural characterization of mitochondrial citrate synthase 4 from Arabidopsis thaliana [Nishio & Mizushima (2020), Acta Cryst. F76, 109-115]. Citrate synthase catalyzes the first reaction in the TCA cycle - a central metabolic pathway that is responsible for the synthesis of several important molecules that provide a major source of cellular energy in plants.
research communications
 access
access

 journal menu
journal menu













![[publBio]](/logos/publbio.gif)





