issue contents
February 2022 issue
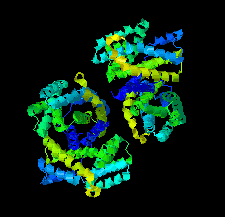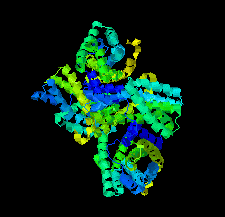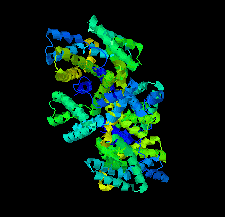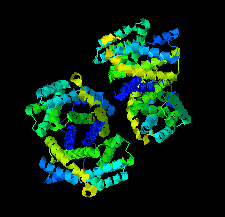
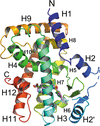
Cover illustration: The urease accessory protein UreF from Klebsiella pneumoniae [Liu et al. (2022), Acta Cryst. F78, 75–80]. Urease is a vital enzyme that is also a virulence factor for K. pneumoniae, an opportunistic pathogen that mostly affects those with weakened immune systems. UreF is an important nickel-binding urease accessory protein involved in the insertion of Ni2+ into the active site of urease.
research communications
 access
access access
access access
access access
accessmethods communications
 access
access

 journal menu
journal menu











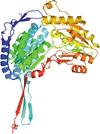




![[publBio]](/logos/publbio.gif)





