issue contents
March 2022 issue
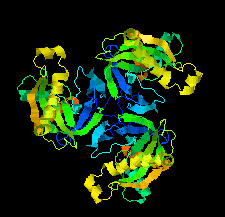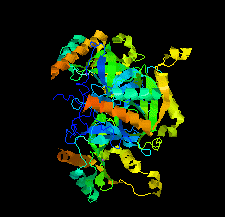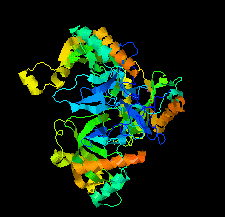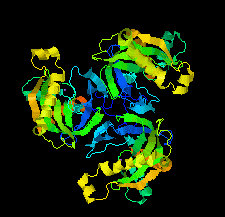
Cover illustration: Chlamydia trachomatis inorganic pyrophosphatase (CtPPase) [Maddy et al. (2022), Acta Cryst. F78, 135–142]. C. trachomatis is the leading cause of bacterial sexually transmitted infections and CtPPase hydrolyzes inorganic pyrophosphate during metabolism. A 2.2 Å resolution X-ray structure of CtPPase reveals shared structural features that may facilitate the repurposing of inhibitors identified for bacterial inorganic pyrophosphatases as starting points for new therapeutics.
topical reviews
 access
accessresearch communications
 access
access access
accessaddenda and errata
 access
access

 journal menu
journal menu















![[publBio]](/logos/publbio.gif)





