issue contents
July 2022 issue
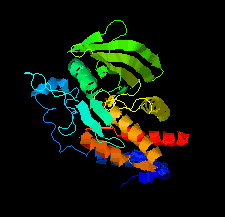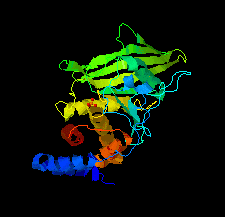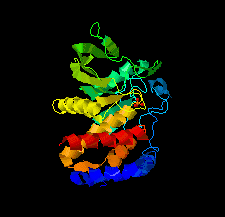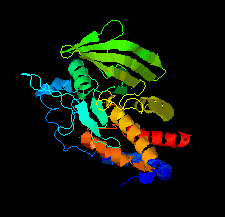
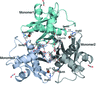
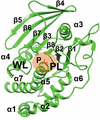
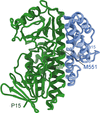
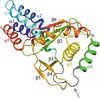
Cover illustration: The catalytic domain of human RPTPH [Kim & Ryu (2022), Acta Cryst. F78, 265–269]. Receptor-type protein tyrosine phosphatases (RPTPs) regulate cell growth, differentiation and death via specific signals, and have been implicated in cancer, diabetes and neurological diseases. RPTPH, a member of the type 3 RPTP family, is an important regulator of colorectal cancer and hepatic carcinoma. Here the crystal structure of the catalytic domain of RPTPH has been determined at 1.56 Å resolution.
research communications
methods communications
 access
access

 journal menu
journal menu












![[publBio]](/logos/publbio.gif)





