issue contents
September 2022 issue

Cover illustration: The crystal structure of acetylcholine-binding protein from Aplysia californica in complex with the naturally occurring neurotoxin (+)-anatoxin-a [Parker et al. (2022), Acta Cryst. F78, 313–323]. Binding assays and crystallography have been used to try to understand the chemical features and interactions that allow two alkaloid neurotoxins to influence human nicotinic acetylcholine receptors, which are important therapeutic targets.
research communications
 access
access access
accessaddenda and errata



 journal menu
journal menu












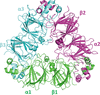
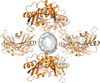


![[publBio]](/logos/publbio.gif)





