issue contents
April 2023 issue
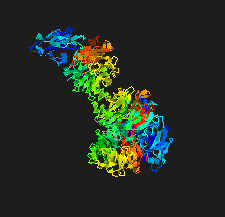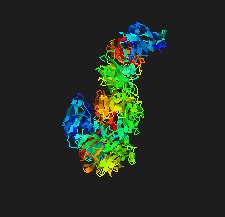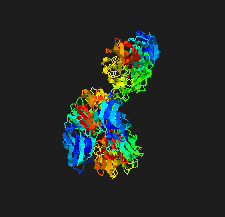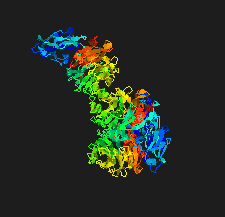
Cover illustration: The crystal structure of the sialidase SiaPG from Porphyromonas gingivalis [Dong et al. (2023), Acta Cryst. F79, 87–94]. P. gingivalis is a pathogenic bacterium which causes human periodontal disease and contributes to bacterial pathogenesis via promoting the formation of biofilms and capsules, and providing nutrients for bacterial colonization. Key residues that are required for substrate binding and catalysis have been identified and structural comparison with other sialidases reveals distinct features of the active-site pocket which might confer substrate specificity. These findings provide the structural basis for the further design and optimization of effective inhibitors to fight against P. gingivalis-derived oral diseases.
introduction
Room-temperature biological crystallography has seen a resurgence in recent years and a collection of articles recently published in
IUCrJ,
Acta Cryst. D Structural Biology and
Acta Cryst. F Structural Biology Communications, have been collected together to produce a virtual special issue at
https://journals.iucr.org/special_issues/2022/RT/.
research communications
The first crystal structure of the widely used vaccine carrier protein CRM197 in its monomeric form reveals features underlying its dimerization and vaccine-conjugation reaction.
The crystal structure of the sialidase SiaPG from Porphyromonas gingivalis, a pathogenic bacterium which causes human periodontal disease, is reported.
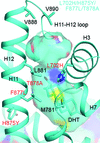
A partially open conformation of the human androgen receptor ligand-binding domain containing four drug-resistance mutations, L702H/H875Y/F877L/T878A, was observed. Of these mutations, both L702H and F877L are important for this conformation, which may affect ligand binding as well as resistance to antagonists.


 access
access
 journal menu
journal menu













![[publBio]](/logos/publbio.gif)





