issue contents
January 2017 issue

editorial
BIOLOGY | MEDICINE
The importance of preserving and making available the original experimental data underlying biological structural models is discussed, both for crystallography, where the raw data images pose particular challenges, and for other structure determination techniques.
scientific commentaries
BIOLOGY | MEDICINE
Macromolecular Big Data provide numerous challenges and a number of initiatives that are starting to overcome these issues are discussed.
BIOLOGY | MEDICINE
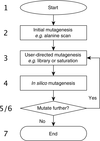
Can in silico engineering speed up the delivery of biocatalysts for the burgeoning bioeconomy? In this issue, Kamerlin and coworkers introduce CADEE [Amrein et al. (2017), IUCrJ, 4, 50–64] – a framework for Computer-Aided Directed Evolution of Enzymes – that promises to lessen the burden on `wet lab' enzymologists when optimizing biocatalysts using laboratory-based directed evolution methods.
research papers
BIOLOGY | MEDICINE
The role of the two tryrosine gating residues in the binding affinity of FimH for substituted mannosides is investigated using a combination of mutagenesis, X-ray crystallography, affinity measurements and molecular simulation. In contrast to the Y48A mutation, it is clearly shown that the Y137A mutation relaxes the binding site prematurely, whereby the stringent selectivity of the FimH lectin for mannose is disrupted and the binding affinity decreases.
PHYSICS | FELS
We present a new method of extracting distributions of orientational order, like bond angles, from fluctuation diffraction data. It has potential applications to study of liquids, glasses and other systems that lack long-range order.
CHEMISTRY | CRYSTENG
Download citation


Download citation


An electron density study on sterically overcrowded octachloronaphthalene is performed using an extensive combination of high-resolution single crystal X-ray diffraction and ab initio single-point and periodic DFT calculations. The unusual asymmetry in the crystalline geometry as opposed to symmetric solvated phase geometry is investigated and the role of steric forces, contributed primarily by the peri interactions in imparting unique `localized double bond' character in the naphthalene ring, is rationalized.
BIOLOGY | MEDICINE
A new computational tool, CADEE (Computer-Aided Directed Evolution of Enzymes), is presented.
CHEMISTRY | CRYSTENG
Download citation


Download citation


Solid-state [2 + 2] photocycloaddition reactivity, mechanical motion during cycloaddition reaction under UV light and naturally curved single crystals are reported in structurally similar mononuclear Zn(II) coordination complexes containing fluoro derivatives of 4-styryl pyridine ligands.
feature articles
NEUTRON | SYNCHROTRON
This article highlights scientific and technical contributions from the Protein Crystallography Station at Los Alamos, the first purpose-built macromolecular crystallography station at a spallation neutron source.
topical reviews
NEUTRON | SYNCHROTRON
There are rapid developments in storage options for the preservation and reuse of raw data within the scientific domain of the IUCr and its Commissions, each of which operates within a great diversity of instrumentation. Formulation of improved metadata descriptors for the raw data is under way. Science policy makers strive towards an `Open Science' model within which crystallographers will increasingly work in the future. Case studies are described that show the community's progress.



 journal menu
journal menu




 access
access