issue contents
March 2018 issue
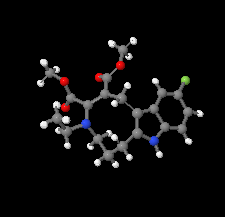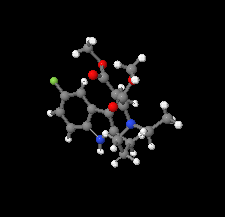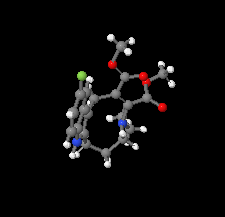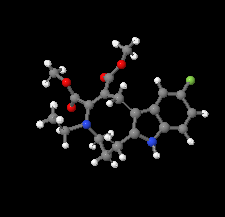
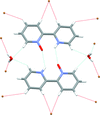
Cover illustration: The azoninoindole dimethyl 4-ethyl-11-fluoro-1,4,5,6,7,8-hexahydroazonino[5,6-b]indole-2,3-dicarboxylate might be considered as a candidate for the design of new Alzheimer drugs. It is the product of a ring-expansion reaction from a seven-membered fluorinated hexahydroazepine to a nine-membered azonine. See: Toze, Listratova, Voskressensky, Chernikova, Lobano, Bilyachenko & Dorovatovskii [Acta Cryst. (2018). E74, 298-301].
research communications
Download citation


Download citation


The title compound comprises a central o-dianisidine unit and terminal benzyl groups. In the molecule, the O atom are involved in intramolecular hydrogen bonding. In the crystal, adjacent molecules are linked by C—H⋯O interactions, forming a one-dimensional ladder along the a axis.
CCDC reference: 1820338
Download citation


Download citation


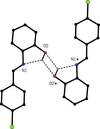
In the title compound, the central OsII ion is ligated by a hexahaptic η6 p-cymene ring, a Br− ligand and two O atoms of a chelating tropolonate group. The p-cymene ligand presents more than one conformation, giving rise to a discrete disorder, which was modelled with two different orientations with occupancy values of 0.561 (15) and 0.439 (15).
CCDC reference: 1818437
Download citation


Download citation


In the title complex, the Cr3+ ion is coordinated by the four O and two N atoms of the 1,3-pdta ligand, displaying a distorted octahedral geometry. The Ag+ cation is surrounded by six O atoms from neighboring 1,3-pdta groups and water molecules.
CCDC reference: 1424813
Download citation


Download citation


3,3′-[(3-Methoxy-4-hydroxyphenyl)methanediyl]bis(4-hydroxy-2H-chromen-2-one), the 4-hydroxy-3-methoxyphenyl-substituted derivative of dicoumarol, was deprotonated by the addition of triethylamine, yielding the respective ammonium salt which was crystallized from a methanol solution. The deprotonated dicoumarol derivative exhibits an intramolecular negative charge-assisted hydrogen bond between the deprotonated and non-deprotonated alcohol functions of the coumarol substituents.
CCDC reference: 1818945
Download citation


Download citation


The crystal structure of the title compound consists of three different negatively charged discrete octahedral iron(III) complexes, that are charge-balanced by 4-methoxypyridinium cations.
CCDC reference: 1821019
Download citation


Download citation


The syntheses of a formazan and a verdazyl radical are reported along with their crystal structures, UV–Vis spectra, and the EPR spectrum of the radical. In addition, the isolation of a possible by-product was achieved.
Download citation


Download citation


The nine-membered azoninoindole dimethyl 4-ethyl-11-fluoro-1,4,5,6,7,8-hexahydroazonino[5,6-b]indole-2,3-dicarboxylate, representing a candidate for the design of new Alzheimer drugs, has been studied by synchrotron X-ray diffraction.
CCDC reference: 1818381
Download citation


Download citation


The coordination geometry in (n-Bu)2Sn[S2CN(CH2CH2OCH3)2]2, (I), is based on a skewed trapezoidal bipyramid, while that in (C6H5)3Sn[S2CN(CH3)CH2CH2OCH3], (II), is based on a tetrahedron. In the crystal of (I), supramolecular layers parallel to (10-1) are sustained by methylene-C—H⋯O(methoxy) interactions, while in (II), centrosymmetric dimers are formed via pairwise weak phenyl-C—H⋯π(phenyl) contacts.
Download citation


Download citation


Anhydrous cobalt(II) chloride reacts with quinoline (C9H7N) in dimethyl sulfoxide (Me2SO) to form the novel complex salt [CoII(Me2SO)6][CoIICl3quinoline]2. The compound comprises an octahedral homoleptic Me2SO-solvated cobalt(II) cation and a tetrahedral cobaltate(II) anion attached to three chloro ligands and one quinoline moiety.
CCDC reference: 1820336
Download citation


Download citation


The packing motifs in the isomeric title compounds feature inversion dimers or chains, mediated by C—H⋯O interactions, one of which is unusually short (H⋯O = 2.18 Å).
Download citation


Download citation


The title compound has enol–imine tautomeric form. E/Z isomerism and enol/keto tautomerism energy barriers have been calculated by relaxed potential energy surface scan calculations with DFT methods.
CCDC reference: 1457124
Download citation


Download citation


The title aroyl hydrazone Schiff base salt, consists of one molecular cation in the keto tautomeric form, adopting an E configuration with respect to the azomethine bond, and one nitrate anion.
CCDC reference: 1822116
Download citation


Download citation


Synthesis and structural studies of a NiIIS4–2{2Fe2S}model for the H-cluster of [FeFe]-hydrogenase.
Download citation


Download citation


The aldoximes (E)-1-naphthaldehyde oxime (I) and E-phenanthrene-9-carbaldehyde oxime (II) were synthesized and characterized using NMR and XRD. The crystal structures of both (I) and (II) are similar with a single intermolecular O—H⋯N hydrogen-bonding interaction, giving rise to the formation of one-dimensional polymeric chains extending along the 21 (b) screw axes in each.
Download citation


Download citation


In the title compound, the two carbazole groups are nearly coplanar, making a dihedral angle of 16.90 (5)°, and are bridged by vinyl. The crystal structure features π–π and C—H⋯π interactions and C—H⋯Br short contacts.
CCDC reference: 1821846
Download citation


Download citation


Structural disorder is observed due to the fact that protonation, as well as oxidation, of the N atoms of 2,2′-bipyridine occurs either at either of the N atoms. The disorder extends to the remainder of the cation, with a refined occupancy rate of 0.717 (4) for the major moiety.
CCDC reference: 1822822
Download citation


Download citation


Even though they contain isosteric and isoelectronic molecules that both form C=O⋯Br contacts, the cyanide and isocyanide crystals are not isomorphous in any dimension.
Download citation


Download citation


In the title compound, a naphthalene derivative, the cyclohexa-1,3-diene ring of the 1,2-dihydronaphthalene ring system adopts a half-chair conformation. The mean plane of the 1,2-dihydronapthalene ring system make dihedral angles of 86.23 (6) and 64.80 (7)° with two phenyl rings. In the crystal, the molecules are linked by C—H⋯O, interactions and further stabilized by C—H⋯π and π–π interactions.
CCDC reference: 1823056
Download citation


Download citation


The molecular structure of (2E)-1,4-bis(4-chlorophenyl)but-2-ene-1,4-dione (1) is composed of two p-chloro phenyl rings, each bonded on opposite ends to a near planar 1,4-trans enedione moiety [–C(= O)—CH=CH—(C=O)–]. (2E)-1,4-Bis(4-bromophenyl)but-2-ene-1,4-dione (2) has a similar structure to (1), but with two p-bromo phenyl rings and a less planar enedione group. In the crystal, molecules of (1) exhibit C—Cl⋯Cl type I interactions, whereas molecules of (2) present C–Br⋯Br type II interactions.
Download citation


Download citation


The crystal structures of two CuII complexes of 1-H-3,4,5-trimethylpyrazole, cis-[{CuCl[3,4,5-(CH3)3(C3H2N2)]2}2(μ-Cl)2] and [Cu{3,4,5-(CH3)3(C3HN2)}4(H2O)](NO3)2, are presented and compared to the 3-methyl-1H-pyrazole and 3,5-dimethyl-1H-pyrozole analogues.
Download citation


Download citation


Crystal structures of two benzothiazinones belonging to a class of compounds implicated in antimicrobial, antitumour, and HIV-RT inhibitory activity are reported.
Download citation


Download citation


In the crystal, N—H⋯N hydrogen bonds link the molecules into the supramolecular chains propagating along the c-axis direction.
CCDC reference: 1820439
Download citation


Download citation


The title compound has a `banana' shape, with the dihedral angle formed by the outer rings being 8.30 (7)°. In the crystal, the three-dimensional architecture features nitrobenzene-C—H⋯O(carbonyl), pyrrole-C—H⋯O(nitro), π(pyrrole)—π(nitrobenzene) and nitro-O⋯π(pyrrole) interactions.
CCDC reference: 1823263
Download citation


Download citation


The molecules of the title compound are almost planar, with an intramolecular O—H⋯N hydrogen bond, and they are linked into a three-dimensional framework by a combination of one O—H⋯O hydrogen bond and two C—H⋯π(arene) hydrogen bonds.
CCDC reference: 1823227
Download citation


Download citation


Different molecular conformations in the crystal structures of three 5-nitroimidazolyl derivatives
The title compounds show different conformations in the solid state.
Download citation


Download citation


The title compound, a cassane-type diterpenoid, was isolated from a medicinally important plant, Caesalpinia pulcherrima (Fabaceae). In the molecule, the three cyclohexane rings are trans-fused and adopt chair, chair and half chair conformations.
CCDC reference: 1823530
Download citation


Download citation


In the structure of an aryltellurenium(II) salt containing a [4-tert-butyl-2,6-bis(1-pentyl-1H-benzimidazol-2-yl)phenyl]tellurium(II) cation and a [HgI3(dioxane)]− anion, the cation and anion are linked by a C—H⋯I interaction.
CCDC reference: 1823822
Download citation


Download citation


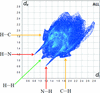
Reaction of N-(4-chlorobenzenesulfonyl)glycinyl hydrazide with 4-nitrobenzaldehyde gives the N,N-dimethylformamide monosolvated N-acylhydrazone derivative, (E)-N-{2-[2-(4-nitrobenzylidene)- hydrazine-1-yl]-2-oxoethyl}-4-χhlorobenzenesulfonamide. Rings of 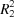 (10) and
(10) and 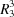 (11) graph-set motifs are formed in the crystal structure by N—H⋯O and C—H⋯O hydrogen bonds. The two-dimensional fingerprint (FP) plots for significant intermolecular interactions indicate that the greatest contribution is from the O⋯H/H⋯O contacts (31.3%), corresponding to N⋯H⋯O/C⋯H⋯O interactions.
(11) graph-set motifs are formed in the crystal structure by N—H⋯O and C—H⋯O hydrogen bonds. The two-dimensional fingerprint (FP) plots for significant intermolecular interactions indicate that the greatest contribution is from the O⋯H/H⋯O contacts (31.3%), corresponding to N⋯H⋯O/C⋯H⋯O interactions.
CCDC reference: 1433601
Download citation


Download citation


The conformations of the two free doxorubicin (DoxH+) cations present in the crystal structure of the title compound and (Dox) bound to proteins and DNA are compared.
CCDC reference: 1815074
Download citation


Download citation


A new NASICON-type phase, Na0.72(Cr0.48,Al1.52)(Mo2.77,Al0.23)O12, was synthesized by solid-state reaction. The structural unit consists of one octahedron M1O6 (M1 = Cr1/Al2) and one tetrahedron M2O4 (M2 = Mo1/Al1) sharing corners. The charge compensation is provided by Na+ cations.
CCDC reference: 1824957
Download citation


Download citation


The title Schiff base compound is considerably non-planar, with the outer phenol and pyridine rings being inclined to each other by 70.21 (3)°.
CCDC reference: 1542988
Download citation


Download citation


The molecule in the title compound approximates a U-shape with the nitrobenzene and ester substituents lying to the same side of the molecule. In the crystal, linear supramolecular chains are sustained by methylene-C—H⋯O(carbonyl) interactions.
CCDC reference: 1825237
Download citation


Download citation


The title compound, an hemisynthetic product, was obtained by the tosylation reaction of the naturally occurring meroterpene p-methoxythymol 1.
CCDC reference: 1825267


 journal menu
journal menu