issue contents
July 2001 issue
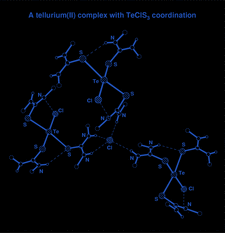
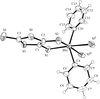
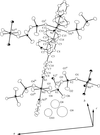
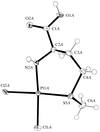
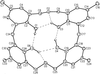
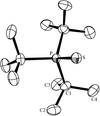
Cover illustration: The molecular packing in chlorotris(N,N'-dicyclohexylthiourea-S)tellurium(II) chloride, with hydrogen bonds indicated by dashed lines. For clarity, only the C atoms (dotted circles) joined to the main structural unit in the cyclohexyl groups are shown. See Husebye, Törnroos & Zhu [Acta Cryst. (2001), C57, 854-856].
inorganic compounds
Download citation


Download citation


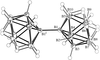
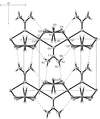
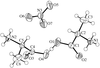
An isomer of B20H26, isolated from the autolysis of nido-B10H14 in a silent electrical discharge, is shown to be the title compound 1,1′-bis(nido-decaboranyl). The molecule has crystallographic inversion symmetry and a long intercage B—B bond of 1.704 (3) Å.
Download citation


Download citation


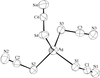
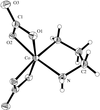
The Ag+ ions in the title compounds are tetrahedrally coordinated by the S atoms of four thiocyanate groups, which act as terminal or 1,1-μ-bridging ligands. The anions in K[Ag(SCN)2] form a two-dimensional polymeric network containing Ag2S2 and Ag6S6 rings.
Download citation


Download citation


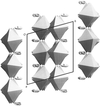
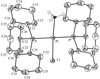
The monoclinic superstructure of Ce-doped SrMgF4 can be described as a stack of perovskite-type slabs in which MgF6 octahedra are rotated in a complicated manner. Sr atoms are shifted from ideal positions to fit in the space formed by the six MgF6 octahedra of one slab and one MgF6 octahedron of a neighbouring slab.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


The title structure consists of [V2O6], [Ni(4,4′-bipy)4O2] and [Ni(H2O)2(4,4′-bipy)2O2] polyhedra, and water of crystallization.
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu