issue contents
October 2012 issue
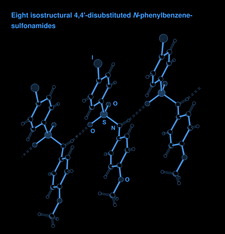
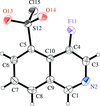
Cover illustration: The N-H O hydrogen-bonded chain in the crystal structure of 4-iodo-N-(4-methoxyphenyl)benzenesulfonamide. Hydrogen bonds are shown as dashed lines. See Gelbrich, Threlfall & Hursthouse [Acta Cryst. (2012), C68, o421-o426].
O hydrogen-bonded chain in the crystal structure of 4-iodo-N-(4-methoxyphenyl)benzenesulfonamide. Hydrogen bonds are shown as dashed lines. See Gelbrich, Threlfall & Hursthouse [Acta Cryst. (2012), C68, o421-o426].
inorganic compounds
Download citation


Download citation


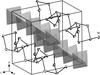
The crystal structure of the cubic compound Ni2Sn2Zn is related to that of Ni5.20Sn8.7Zn4.16Cu1.04 and shows common structural elements with the atomic arrangements of the high-temperature phase of Ni3Sn2 and with Ni5Sn4Zn. It exhibits a considerable homogeneity range, along with mixed occupations and partially occupied sites.
Download citation


Download citation


The title compound, [Te8][NbOCl4]2, crystallizes with triclinic lattice symmetry and consists of infinite {[Te8]2+}n cations associated with pyramidal [NbOCl4]− anions. The novel catena-octatellurium(2+) cation is composed of Te5 rings that are linked via Te3 units.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu