issue contents
September 2016 issue
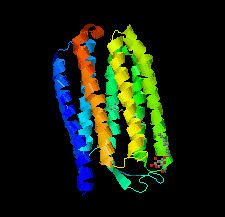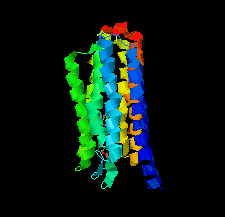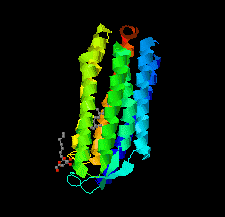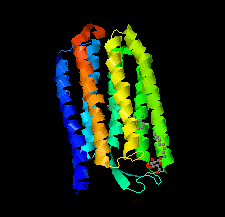
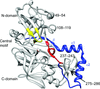
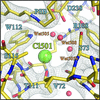
Cover illustration: Halobacterium salinarum halorhodopsin with a partially depopulated primary chloride-binding site (Schreiner et al., p. 692). Halophilic archaea such as H. salinarum thrive in highly saturated brines. In order to maintain the osmotic balance, the transmembrane pump halorhodopsin translocates chloride ions from the extracellular to the cytoplasmic side upon illumination. Here, to further elucidate this ion-transport mechanism at the molecular level, crystals have been treated to provoke the loss of chloride.
research communications
 access
access access
access access
access

 journal menu
journal menu

















![[publBio]](/logos/publbio.gif)





