issue contents
December 2016 issue
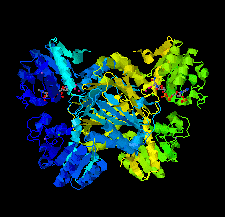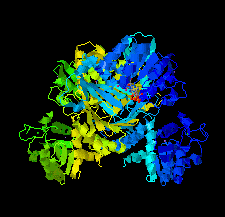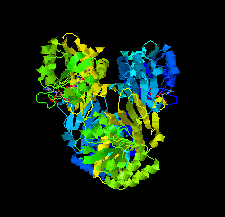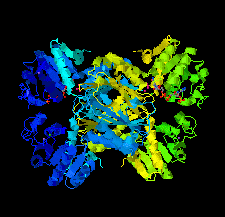
Cover illustration: The crystal structure of dihydrodipicolinate reductase from the human-pathogenic bacterium Bartonella henselae strain Houston-1 at 2.3 Å resolution (Cala et al., p. 885).
research communications
The structure of the A. gambiae serine proteinase inhibitor (serpin) SRPN18 was determined, revealing an unusually short and constricted reactive-center loop.
PDB reference: SRPN18 from Anopheles gambiae, 5c98
The crystal structure of Msmeg_6760 reveals a novel fold for a toxin that harbors a large central cavity that is likely to bind a large hydrophobic compound.
PDB reference: Msmeg_6760, 3uid
Open  access
access
 access
accessA cellulase from K. pneumoniae was cloned, expressed, purified and shown to be an endoglucanase. Its structure was determined at 1.76 Å resolution and comparisons with homologous enzymes provide information on substrate specificity.
PDB reference: Cel10, 5gy3
The expression, purification, crystallization, X-ray powder diffraction data collection and preliminary analysis of protease 3C from coxsackievirus B3 are reported.
The cloning, expression, purification, crystallization and X-ray diffraction analysis of dihydrodipicolinate reductase from the human-pathogenic bacterium B. henselae, the causative bacterium of cat-scratch disease, are reported.
PDB reference: dihydrodipicolinate reductase from B. henselae, 3ijp
Open  access
access
 access
accessFapF from Pseudomonas facilitates the secretion of the amyloid-forming polypeptide FapC across the bacterial outer membrane. A protocol for forming reproducible well diffracting crystals is described in which the switch to tetraethylene glycol monooctyl ether detergent during purification proved to be critical and represents a handy tip for β-barrel protein crystallography.
A hemihedral twinning problem was found in crystals of a tetrahydrofolate-dependent O-demethylase, LigM, from Sphingobium sp. SYK-6. The problem was overcome using microseeding.
The high-resolution (1.57 Å) structure and kinetic data of the key fire blight phosphatase AmsI are reported.
PDB reference: AmsI, 4d74
addenda and errata
Free 



 journal menu
journal menu

















![[publBio]](/logos/publbio.gif)






