issue contents
April 2018 issue
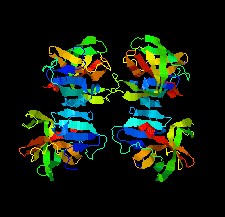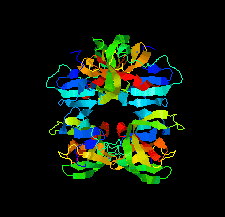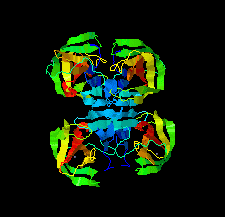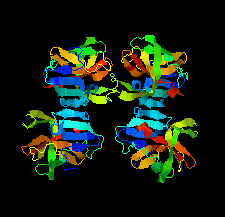
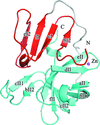
Cover illustration: The structure of 2A protease from human rhinovirus C15 (Ling et al., p. 255). The majority of outbreaks of the common cold are caused by human rhinoviruses (HRVs), and the 2A protease of HRVs is known to play an important role in the propagation of the virus and in viral replication. It is hoped that this structure will help in the design of efficient peptide inhibitors or antiviral compounds to block the replication of HRVs.
research communications
Structural view of the helicase reveals that Zika virus uses a conserved mechanism for unwinding RNA
 access
access

 journal menu
journal menu

















![[publBio]](/logos/publbio.gif)





