issue contents
April 2019 issue
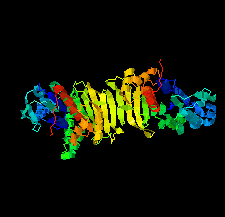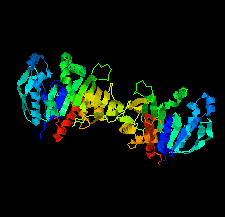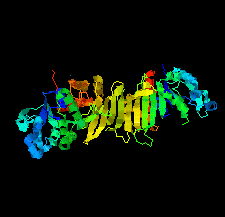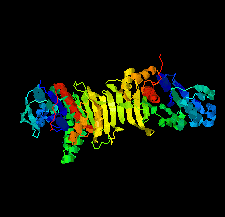
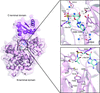
Cover illustration: Phosphoribulokinase (PRK) from Synechococcus sp. strain PCC 6301 [Wilson et al. (2019), Acta Cryst. F75, 278-289]. PRK catalyses the ATP-dependent phosphorylation of ribulose 5-phosphate to give ribulose 1,5-bisphosphate and regulation of this reaction in response to light controls carbon fixation during photosynthesis. Here, the crystal structure of PRK shows that the enzyme is dimeric and has an ![[alpha]](/logos/entities/alpha_rmgif.gif) /
/![[beta]](/logos/entities/beta_rmgif.gif) -fold with an 18-stranded
-fold with an 18-stranded ![[beta]](/logos/entities/beta_rmgif.gif) -sheet at its core. Interestingly, a disulfide bond is found between Cys40 and the P-loop residue Cys18, revealing the structural basis for the redox inactivation of PRK activity. A second disulfide bond appears to rigidify the dimer interface and may thereby contribute to regulation by the adaptor protein CP12 and glyceraldehyde-3-phosphate dehydrogenase.
-sheet at its core. Interestingly, a disulfide bond is found between Cys40 and the P-loop residue Cys18, revealing the structural basis for the redox inactivation of PRK activity. A second disulfide bond appears to rigidify the dimer interface and may thereby contribute to regulation by the adaptor protein CP12 and glyceraldehyde-3-phosphate dehydrogenase.
research communications
 access
access access
access access
access

 journal menu
journal menu











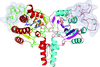








![[publBio]](/logos/publbio.gif)





