issue contents
November 2019 issue
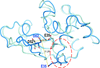
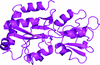
Cover illustration: The crystal structure of the P235GK mutant of the arginine-binding protein from Thermotoga maritima in its monomeric state provides clues to the structural determinants of the domain swapping observed in the wild-type protein [Smaldone et al. (2019), Acta Cryst. F75, 707-713]. The data analysis, solution, refinement and validation of this structure were conducted as part of a tutorial session during the Crystallographic Information Fiesta held in Naples, Italy in the summer of 2019 and organized by the Italian Crystallographic Association in partnership with the IUCr.
editorial



 journal menu
journal menu













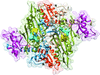
![[publBio]](/logos/publbio.gif)





