issue contents
August 2020 issue
Cover illustration: One of three structures of closely related NTF2-like superfamily proteins determined by Vuksanovic et al. [(2020), Acta Cryst. F76, 372-383]. The presence of a solvent-accessible cavity and the conservation of the His/Asp dyad suggest a potential enzymatic role for these enzymes in polyketide biosynthesis.
research communications
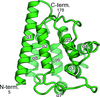
FYCO1 plays an important role in the kinesin-dependent microtubule plus-end-directed transport of autophagosomes. Here, the crystal structure of the RUN domain of FYCO1 has been determined.
PDB reference: FYCO1 RUN domain, 7bqi
Open  access
access
 access
accessThe structure of the FAD-containing ferredoxin reductase FdR9 from Thermobifida fusca was obtained at 1.9 Å resolution.
PDB reference: Fdr9, 6tuk
The crystal structure of GH30-7 endoxylanase C from the filamentous fungus Talaromyces cellulolyticus was determined at 1.65 Å resolution. Structural and mutational analyses revealed that subsite −2b possesses unique structural features that interact with arabinofuranosyl and 4-O-methyl-α-D-glucuronosyl side chains in a xylan substrate.
PDB reference: GH30-7 endoxylanase C, 6m5z
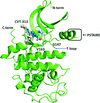
The binding of CVT-313 to cyclin-dependent kinase 2 (CDK2) was confirmed using a thermal shift assay and its cellular potency was demonstrated in a CTG-based cell-proliferation assay. The crystal structure of the CDK2–CVT-313 complex was solved to a resolution of 1.74 Å. The structural information obtained from this study is expected to facilitate the design of more potent and selective CDK2 inhibitors for the treatment of cancer.
PDB reference: CDK2–CVT-313 complex, 6inl
Open  access
access
 access
accessThe structure of the uncomplexed Rab-binding domain of Rab11 family-interacting protein 2 has been determined at 2.3 Å resolution. The structure reveals antiparallel α-helical dimers that associate in the crystal lattice through polar interactions. The dimers assemble into a four-helix bundle in the lattice that is brought together by a remarkable stack of arginine residues.
PDB reference: Rab11 family-interacting protein 2, 6s8x
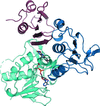
The crystal structure of the aminoglycoside 6′-N-acetyltransferase from Enterococcus faecium is reported in a novel substrate-free form in order to understand the mechanism underlying its substrate recognition in detail. It is proposed that the enzyme sequentially undergoes conformational selection and induced fit for substrate binding.
PDB reference: aminoglycoside 6′-N-acetyltransferase, 7bxz
The structures of three closely related NTF2-like superfamily proteins that are involved in type II polyketide biosynthesis in as-yet-unknown ways were determined. The presence of a solvent-accessible cavity and the conservation of the His/Asp dyad suggest a potential enzymatic role for these enzymes in polyketide biosynthesis.
The crystal structure of the extracellular domain of TMK3 shows that the non-leucine-rich-repeat region plays an essential role in its integrity.
PDB reference: extracellular domain of TMK3, 7brc


 journal menu
journal menu














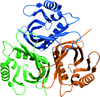

![[publBio]](/logos/publbio.gif)





