issue contents
March 2021 issue
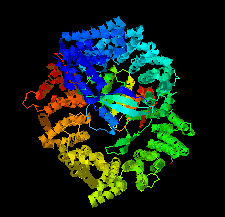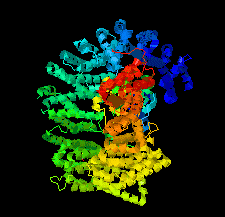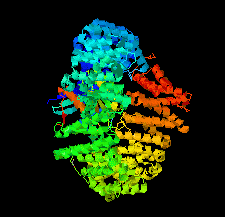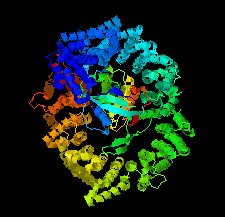
Cover illustration: Human CRM1, covalently modified by 2-mercaptoethanol on Cys528, in complex with RanGTP [Shaikhqasem et al. (2021), Acta Cryst. F77, 70-78]. CRM1 is a nuclear export receptor that has been important in the development of antitumor and antiviral drugs. Structural analysis of several inhibitor compounds bound to CRM1 revealed that their mechanism of action relies on the covalent modification of a critical cysteine residue (Cys528 in the human receptor) located in the nuclear export signal-binding cleft.
research communications
 access
access

 journal menu
journal menu











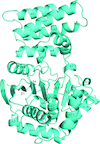



![[publBio]](/logos/publbio.gif)





