issue contents
June 2021 issue
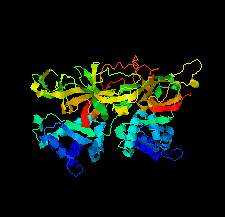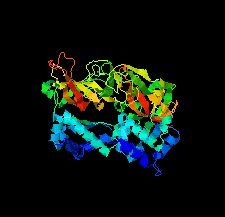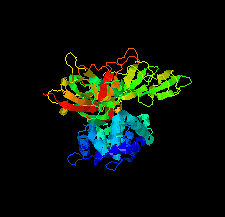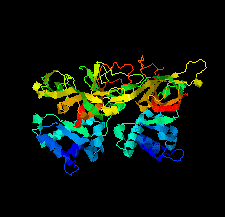
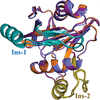
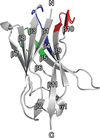
Cover illustration: The minichromosome maintenance (MCM) complex forms a hexameric ring that unwinds DNA at the replication forks of all eukaryotes and archaea. The crystal structure of a dimer of the N-terminal domain of MCM from Sulfolobus solfataricus [Meagher et al. (2021), Acta Cryst. F77, 177–186] suggests a straightforward model for the action of the MCM ring.
research communications




 journal menu
journal menu











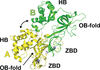

![[publBio]](/logos/publbio.gif)





