issue contents
July 2021 issue
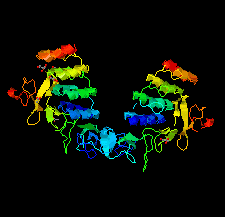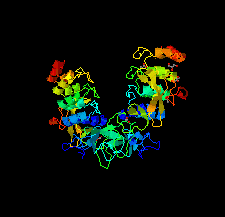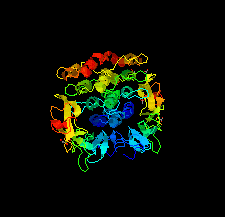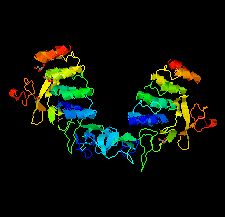
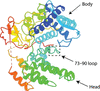
Cover illustration: HER3 extracellular domain 4 in complex with the designed ankyrin-repeat protein D5 [Radom et al. (2021), Acta Cryst. F77, 192–201]. The members of the human epidermal growth factor receptor (HER) family are among the most intensely studied oncological targets. HER3 is a key oncogene, regulating the activity of other receptors and being involved in progression and tumor escape in multiple types of cancer. Designed ankyrin-repeat proteins (DARPins) serve as antibody mimetics that have proven to be useful in the clinic, in diagnostics and in research. Here, the selection of DARPins targeting extracellular domain 4 of HER3 is described.
research communications
 access
access access
access access
access access
access
 journal menu
journal menu














![[publBio]](/logos/publbio.gif)





